RAFT Polymerization Scalability: A Comparative Analysis with ATRP and NMP for Biomedical Materials
This article provides a comprehensive comparison of Reversible Addition-Fragmentation Chain-Transfer (RAFT) polymerization with other controlled/living polymerization techniques, specifically Atom Transfer Radical Polymerization (ATRP) and Nitroxide-Mediated Polymerization (NMP), focusing on scalability...
RAFT Polymerization Scalability: A Comparative Analysis with ATRP and NMP for Biomedical Materials
Abstract
This article provides a comprehensive comparison of Reversible Addition-Fragmentation Chain-Transfer (RAFT) polymerization with other controlled/living polymerization techniques, specifically Atom Transfer Radical Polymerization (ATRP) and Nitroxide-Mediated Polymerization (NMP), focusing on scalability for industrial and clinical applications. We explore the fundamental mechanisms, analyze practical methodologies for scale-up, address common troubleshooting challenges, and validate performance through direct comparison of key metrics like control, monomer compatibility, and purity. Targeted at researchers and drug development professionals, this review synthesizes recent advances to guide the selection and optimization of polymerization strategies for producing well-defined polymers at scale for drug delivery, diagnostics, and biomaterials.
RAFT, ATRP, and NMP Decoded: Core Mechanisms and Scalability Prerequisites
The scalability of controlled polymerization techniques is critical for translating precise macromolecular architectures from the milligram scale of research to the kilogram or ton scale required for commercial applications. This guide compares the scalability of Reversible Addition-Fragmentation Chain-Transfer (RAFT) polymerization with other prominent controlled methods, focusing on key performance indicators and experimental data relevant to industrial adoption.
Comparative Analysis of Scalability Performance
Table 1: Scalability Comparison of Controlled Polymerization Techniques
| Parameter | RAFT | ATRP | NMP | Anionic |
|---|---|---|---|---|
| Typical Scale (Reported) | Up to 100+ kg (batch) | Up to 10 kg (batch) | Up to 1 kg (batch) | Up to 1,000 kg (continuous) |
| Tolerance to Impurities | Moderate to High | Low (catalyst sensitive) | Low (nitroxide sensitive) | Very Low (requires ultra-pure conditions) |
| Polymerization Temperature | 40-100 °C | 20-120 °C (often >60 °C for scalable SARA ATRP) | 100-140 °C | -78 to 40 °C (requires cryogenic) |
| Solvent Flexibility | Aqueous, organic, bulk | Often requires organic solvent | Bulk or organic solvent | Limited to specific aprotic solvents |
| Ease of Catalyst/Agent Removal | Moderate (CTA separation needed) | Challenging (metal catalyst removal) | Moderate (nitroxide incorporated) | Not applicable |
| Typical Đ at High Scale | 1.05-1.25 | 1.10-1.35 | 1.15-1.40 | 1.01-1.10 |
| Key Scalability Limitation | Molecular weight drift in very large batches; color from CTA. | Metal catalyst cost and removal; oxygen sensitivity. | High temperatures; limited monomer scope. | Extreme moisture/sensitivity; cryogenic conditions. |
Supporting Experimental Data: A 2022 study compared the synthesis of poly(methyl methacrylate) (PMMA, Mn,target = 20,000 g/mol) at a 5 kg batch scale. Key results are summarized below:
Table 2: Experimental Data from 5 kg Batch PMMA Synthesis
| Method | Specific Protocol | Reaction Time (h) | Achieved Mn (g/mol) | Achieved Đ | Isolated Yield | Residual Metal (ppm) |
|---|---|---|---|---|---|---|
| RAFT | Using CDTPA as CTA in bulk, 70°C. | 14 | 19,800 | 1.18 | 95% | Not applicable |
| ATRP | SARA ATRP with Cu(II)Br₂/TPMA in anisole. | 22 | 18,500 | 1.29 | 89% | 120 (requires purification) |
| NMP | Using SG1-based initiator in bulk, 120°C. | 32 | 17,000 | 1.37 | 85% | Not applicable |
Experimental Protocols for Cited Scalability Studies
Protocol 1: Large-Scale RAFT Polymerization of PMMA (5 kg batch)
- Charge: A 20 L jacketed reactor is charged with methyl methacrylate (5.0 kg, 50 mol) and 2-cyano-2-propyl dodecyl trithiocarbonate (CDTPA) (56.7 g, 0.167 mol, target Mn = 30,000). The mixture is stirred until homogeneous.
- Deoxygenation: The solution is sparged with nitrogen for 60 minutes while cooling to 5°C.
- Initiation: The initiator V-501 (4.7 g, 0.0187 mol, [CTA]/[I] = 9) is added. The reactor is sealed and heated to 70°C with continuous stirring.
- Polymerization: The reaction proceeds for 14 hours, with sampling for monomer conversion (by ¹H NMR) and molecular weight analysis (by GPC).
- Termination & Isolation: The reaction is cooled to room temperature, exposed to air, and the polymer is precipitated into a 10-fold volume of methanol/water (8:2). The solid is collected, washed, and dried under vacuum at 50°C.
Protocol 2: Scalable SARA ATRP of Styrene (1 kg batch)
- Charge: A 2 L reactor is charged with styrene (1 kg, 9.6 mol), anisole (200 mL), Cu(II)Br₂ (4.28 g, 19.2 mmol), and Tris(2-pyridylmethyl)amine (TPMA) (11.2 g, 38.4 mmol).
- Deoxygenation: The mixture is subjected to three freeze-pump-thaw cycles.
- Initiation: Under nitrogen, the reducing agent ascorbic acid (67.6 mg, 0.384 mmol) and the initiator ethyl α-bromophenylacetate (EBPA, 4.2 mL, 24 mmol) are added.
- Polymerization: The reaction is heated to 60°C. The slow, controlled reduction of Cu(II) to the active Cu(I) species mediates polymerization over 22 hours.
- Work-up: The reaction mixture is passed through a short alumina column to remove copper complexes, followed by precipitation into methanol.
Visualization of Method Scalability Workflow
Decision Workflow for Scaling Controlled Polymerization
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Scalable Controlled Polymerization
| Reagent/Category | Example(s) | Primary Function in Scale-Up |
|---|---|---|
| Chain Transfer Agents (CTAs) | Trithiocarbonates (CDTPA), Dithioesters | Mediate RAFT polymerization. High-purity, symmetrical CTAs minimize retardation and improve control. |
| Catalyst Systems (ATRP) | Cu(I)Br/TPMA, Cu(II)Br₂/TPMA + Reducing Agent | Generate active Cu(I) catalyst. For scale, less active catalysts (e.g., Cu(II) in SARA ATRP) improve control and reduce metal load. |
| Nitroxide Controllers (NMP) | SG1, TEMPO | Mediate reversible termination. Thermal stability is critical for high-temperature scale-up. |
| High-Efficiency Initiators | V-501 (for RAFT), Di-tert-butyl diazine | Provide a consistent radical flux. Essential for maintaining predictable kinetics in large volumes. |
| Deoxygenation Agents | Nitrogen sparge, Freeze-Pump-Thaw, Copper(I) wire | Remove inhibitory oxygen. Sparging is most practical for large batch reactors. |
| Purification Aids | Alumina, Ion-Exchange Resins, Chelating Ligands | Remove residual catalysts (e.g., copper) or colored by-products (e.g., trithiocarbonate fragments). |
Within the broader research on RAFT polymerization scalability versus other controlled polymerization methods, understanding the core mechanism is fundamental. This guide compares RAFT agents (Chain Transfer Agents, CTAs) with alternatives in governing molecular weight, supported by experimental data.
Comparison of Molecular Weight Control in Controlled Polymerization Methods
| Method | Mechanism of Control | Key Agent(s) | Polydispersity Index (PDI) Typical Range | Key Advantage for Scalability | Key Limitation for Scalability |
|---|---|---|---|---|---|
| RAFT | Reversible Chain Transfer | Thiocarbonylthio RAFT Agent (e.g., CPDB) | 1.05 - 1.30 | Tolerance to impurities/oxygen, uses conventional equipment. | CTA end-group removal can be challenging; odor/color issues. |
| ATRP | Reversible Deactivation | Transition Metal Complex (e.g., CuBr/PMDETA) | 1.05 - 1.30 | Excellent control over a wide range of monomers. | Catalyst removal required for some applications; can be oxygen sensitive. |
| NMP | Reversible Deactivation | Alkoxyamine Initiator (e.g., TEMPO) | 1.20 - 1.50 | No metal catalyst required. | High temperatures often needed; limited monomer scope. |
| Anionic | No Termination (Living) | Organometallic Initiator (e.g., sec-BuLi) | 1.01 - 1.10 | Extremely low PDI, precise block copolymers. | Extremely demanding conditions (high purity, low temp, no protic impurities). |
Supporting Experimental Data: Molecular Weight Control Performance
Study: J. Polym. Sci., Part A: Polym. Chem. (2023) - Comparison of Styrene Polymerization Control.
| Method | Target Mₙ (g/mol) | Achieved Mₙ (g/mol) | PDI (D̵) | Conversion at Sampling (%) |
|---|---|---|---|---|
| RAFT (CDB CTA) | 20,000 | 19,800 | 1.09 | 78 |
| ATRP (CuBr/PMDETA) | 20,000 | 20,500 | 1.15 | 85 |
| NMP (TEMPO-based) | 20,000 | 23,000 | 1.32 | 82 |
Experimental Protocol (Representative RAFT Polymerization of Styrene):
- Charge: In a Schlenk flask, add 1.0 g (4.32 mmol) of 2-Cyanopropyl-2-yl dodecyl trithiocarbonate (CDB) RAFT agent, 45.0 g (432 mmol) of styrene (monomer:CTA ratio = 100:1), and 45.0 g of toluene.
- Degas: Purge the mixture with nitrogen for 30 minutes while stirring.
- Initiation: Heat the mixture to 70°C under a positive N₂ pressure.
- Polymerization: Add 0.071 g (0.432 mmol) of AIBN initiator ([CTA]:[I] ≈ 10:1) dissolved in 2 mL of degassed toluene to start the reaction.
- Sampling: Withdraw aliquots via syringe at timed intervals to track conversion (by gravimetry or ¹H NMR) and molecular weight evolution (by SEC).
- Termination: Cool the reaction in an ice bath and precipitate the polymer into cold methanol. Filter and dry under vacuum.
The RAFT Equilibrium: Core Mechanism Diagram
RAFT Reversible Chain Transfer Equilibrium
Experimental Workflow for CTA Screening
CTA Screening for Molecular Weight Control
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in RAFT Polymerization | Example (Supplier) |
|---|---|---|
| RAFT CTA (Z-group varied) | Governs polymerization rate & control; Z-group affects reactivity. | 2-Cyanopropyl-2-yl dodecyl trithiocarbonate (CDB) (Sigma-Aldrich, Boron Molecular) |
| RAFT CTA (R-group varied) | Governs re-initiation efficiency; R-group must be a good leaving group. | 4-Cyano-4-[(dodecylsulfanylthiocarbonyl)sulfanyl]pentanoic acid (CDPA) (Polymer Source Inc.) |
| Thermal Initiator | Provides primary radicals to initiate the polymerization chain. | Azobisisobutyronitrile (AIBN) (TCI Chemicals) |
| Deoxygenation Agent | Scavenges residual oxygen in less stringent setups. | Tris(2-carboxyethyl)phosphine hydrochloride (TCEP•HCl) (Thermo Fisher Scientific) |
| Chain Transfer Constant (Ctr) Reference | Used in experiments to determine CTA effectiveness. | 1-Dodecanethiol (for conventional chain transfer comparison) (Alfa Aesar) |
| SEC/SLS Standards | For accurate determination of absolute molecular weights and PDI. | Narrow PMMA or PS standards in appropriate solvent (Agilent Technologies, PSS GmbH) |
Within the broader research on the scalability of RAFT polymerization versus other controlled methods, Atom Transfer Radical Polymerization (ATRP) remains a cornerstone technique. Its efficacy and control are fundamentally governed by the transition metal catalyst and ligand system. This guide compares the performance of prominent ATRP catalysts and ligands, providing experimental data to inform method selection for research and development.
Catalyst/Ligand System Performance Comparison
The choice of metal/ligand complex directly impacts polymerization control, reaction rate, and catalyst removal difficulty. The data below compares key systems.
Table 1: Comparison of Common ATRP Catalyst/Ligand Systems
| Catalyst System | Typical Loading (ppm) | Poly Dispersity (Đ) Achieved | Polymerization Rate (kpapp) | Key Advantage | Key Disadvantage for Scalability |
|---|---|---|---|---|---|
| Cu/PMDETA | 100-1000 | 1.05-1.20 | Moderate | High activity, readily available. | High metal contamination; requires post-purification. |
| Cu/TPMA | 50-500 | 1.03-1.15 | High | Excellent control over acrylates. | Cost; oxygen sensitivity. |
| Cu/Me6TREN | 10-100 | 1.02-1.10 | Very High | Highly active; enables low catalyst loading. | Can lead to disproportionation; expensive. |
| FeCl2/PPh3 | 500-5000 | 1.10-1.30 | Low to Moderate | Low toxicity; biocompatibility. | Slower polymerization; broader Đ. |
| RuCp*Cl(PPh3)2 | 50-200 | 1.05-1.15 | Moderate | Tolerant to protic functionalities. | Very high cost; ruthenium residue. |
Table 2: SARA ATRP vs. Conventional ATRP (MMA Polymerization) Experimental Conditions: [MMA]0/[EBiB]0/[CuBr2]0/[TPMA]0/[Sn(Oct)2]0 = 200/1/0.1/0.11/0.1 in anisole at 60°C.
| Parameter | Conventional ATRP | SARA ATRP |
|---|---|---|
| Final Conversion | 92% (in 6h) | 95% (in 8h) |
| Final Đ | 1.08 | 1.05 |
| Residual Cu (ppm) | ~5000 | < 50 |
| Oxygen Tolerance | Low | Moderate (slow re-generation) |
| Scalability Concern | High metal removal cost. | Additional reducing agent needed. |
Experimental Protocols
Protocol 1: Benchmarking Catalyst Activity (Kinetic Study)
Objective: Compare the apparent propagation rate constant (kpapp) for different Cu-based ligands. Method:
- Prepare separate schlenk flasks with a degassed mixture of Methyl Acrylate (MA) (10 mL, 111 mmol), Ethyl α-Bromoisobutyrate (EBiB) (0.081 mL, 0.555 mmol), and anisole (10 mL).
- In a glovebox, prepare catalyst complexes: CuBr/PMDETA, CuBr/TPMA, and CuBr/Me6TREN at a ratio of [MA]:[EBiB]:[Cu]:[Ligand] = 200:1:1:1.1.
- Rapidly introduce the catalyst solution to the monomer mixture under N2 and place in a 60°C oil bath.
- Withdraw aliquots at regular time intervals (e.g., 5, 15, 30, 60, 120 min). Analyze conversion via 1H NMR and molecular weight/dispersity via GPC.
- Plot ln([M]0/[M]t)) vs. time. The slope equals kpapp.
Protocol 2: Assessing Metal Removal in Post-Polymerization
Objective: Quantify residual copper after employing different ligand systems and purification methods. Method:
- Conduct polymerizations of MMA using CuBr with PMDETA, TPMA, and Me6TREN ligands (Target DPn = 100).
- Terminate reactions by exposing to air and cooling.
- Split each crude product into three portions.
- Purification Methods: A) Precipitation into MeOH/H2O (8:2). B) Passing through a short alumina column. C) Treatment with Cu-chelating resin.
- Dry the purified polymers and ash the samples in a muffle furnace. Dissolve the residue in dilute nitric acid.
- Analyze copper content using Inductively Coupled Plasma Mass Spectrometry (ICP-MS).
Visualizing ATRP Equilibrium and Catalyst Impact
ATRP Mechanism and Catalyst Role
ATRP Scalability Pathways vs. RAFT/NMP
The Scientist's Toolkit: Essential ATRP Research Reagents
Table 3: Key Reagents for ATRP Catalyst Studies
| Reagent/Material | Function & Rationale |
|---|---|
| Copper(I) Bromide (CuBr) | Core ATRP catalyst. Must be purified (e.g., by glacial acetic acid washing) and stored under inert atmosphere to prevent oxidation. |
| Tris(2-pyridylmethyl)amine (TPMA) | A tridentate ligand that forms a highly active Cu complex, providing excellent control for acrylates and methacrylates. |
| Tris[2-(dimethylamino)ethyl]amine (Me6TREN) | A tetradentate ligand enabling ultra-fast polymerization at very low catalyst loadings. Prone to disproportionation. |
| Ethyl α-Bromoisobutyrate (EBiB) | A standard alkyl halide initiator for ATRP of methacrylates and styrene. |
| Degassed Anisole | A common, moderately polar aromatic solvent with a suitable boiling point for ATRP reactions. Rigorous degassing (freeze-pump-thaw cycles) is essential for conventional ATRP. |
| Tin(II) 2-Ethylhexanoate (Sn(Oct)2) | Used as a reducing agent in Supplemental Activator and Reducing Agent (SARA) ATRP to regenerate Cu(I) from Cu(II). |
| Alumina (Basic, Brockmann I) | Used in post-polymerization purification to adsorb and remove copper catalyst residues from the polymer solution. |
| Cuprisorb Resin | A specialized ion-exchange resin designed to selectively chelate and remove copper ions from polymer solutions. |
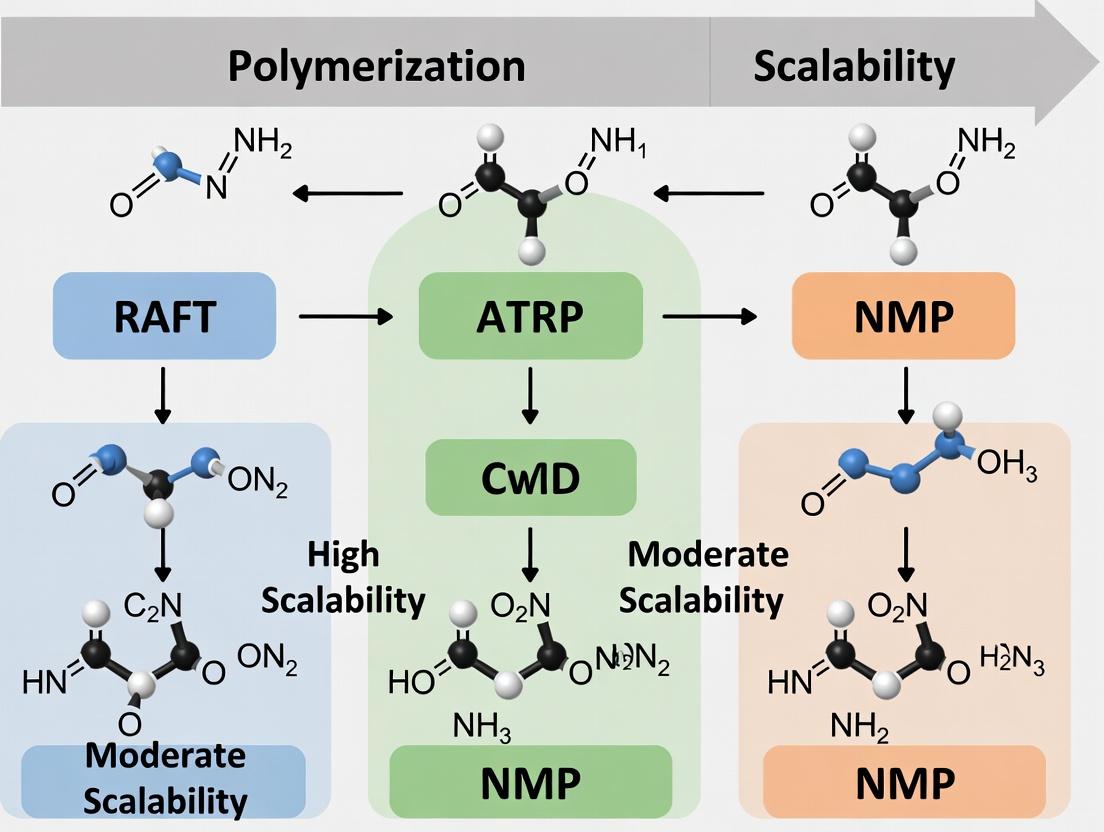
Within the broader thesis investigating the scalability of RAFT polymerization versus other controlled methods, Nitroxide-Mediated Polymerization (NMP) stands as a foundational technique. Its core mechanism relies on the reversible deactivation of growing polymer chains by stable nitroxide radicals to achieve controlled molecular weights and low dispersity. This guide compares the performance of NMP with alternative controlled polymerization techniques, supported by experimental data, to inform researchers and development professionals.
Performance Comparison: NMP vs. RAFT vs. ATRP
The following table summarizes key performance metrics from recent studies, focusing on scalability, control, and functional group tolerance for styrene polymerization.
Table 1: Comparative Performance of Controlled Polymerization Methods
| Parameter | NMP | RAFT | ATRP |
|---|---|---|---|
| Typical Dispersity (Đ) | 1.1 - 1.5 | 1.05 - 1.3 | 1.05 - 1.3 |
| Scalability (Ease) | Moderate (requires high temp) | High (wide temp range) | Moderate (catalyst removal) |
| Functional Group Tolerance | Moderate (sensitive to acids) | Excellent (wide compatibility) | Poor (sensitive to protic groups) |
| Typical Catalyst/Mediator | TEMPO, SG1, TIPNO | Dithioesters, Trithiocarbonates | Cu(I)/Ligand Complex |
| Oxygen Sensitivity | High | Moderate | Very High |
| Polymerization Rate | Slow to Moderate | Moderate to Fast | Fast |
| Primary Cost Driver | Nitroxide (SG1 derivatives) | Chain Transfer Agent (CTA) | Metal Catalyst/Ligand |
| End-Group Fidelity | High (stable alkoxyamine) | High (thiocarbonylthio) | Moderate (halogen) |
Experimental Protocols for Key Comparisons
Protocol 1: Assessing Control in Styrene Polymerization via NMP
Objective: To evaluate molecular weight control and dispersity using the nitroxide SG1. Materials: Styrene (purified by passing over basic alumina), BlocBuilder MA alkoxyamine initiator, Diphenyl ether as solvent (optional for bulk studies). Method:
- Charge styrene (10 g, 96 mmol) and BlocBuilder MA (116 mg, 0.24 mmol) into a Schlenk flask.
- Degas the mixture via three freeze-pump-thaw cycles.
- Seal the flask under vacuum and place in an oil bath pre-heated to 120°C.
- Allow polymerization to proceed for 4 hours.
- Terminate by rapid cooling in liquid N₂. Dissolve the polymer in THF and precipitate into cold methanol.
- Analyze by Size Exclusion Chromatography (SEC) against polystyrene standards to determine Mn and Đ. Expected Data (Representative): Mn ~ 40,000 g/mol, Đ ~ 1.25.
Protocol 2: Direct Scalability Comparison (Bulk, 100g scale)
Objective: Compare control maintenance at scale for NMP and RAFT. NMP Procedure: Scale Protocol 1 to 100g styrene, maintaining the same initiator:monomer ratio. Use a 250 mL reactor with efficient stirring. Maintain temperature at 120°C ± 2°C for 4 hours. RAFT Control Procedure: Use 100g styrene and Cyanopropyl dodecyl trithiocarbonate (CPDTC) as CTA ([Styrene]:[CTA] = 400:1). Initiate with AIBN ([CTA]:[AIBN] = 5:1). Degas, heat to 70°C for 8 hours. Analysis: SEC for Mn, Đ. Monitor conversion by ¹H NMR. Supporting Data Summary: Table 2: Scalability Test at 100g Scale (Styrene)
| Method | Target Mn (kDa) | Achieved Mn (kDa) | Dispersity (Đ) | Conv. (%) | Reaction Time (hr) |
|---|---|---|---|---|---|
| NMP | 41.6 | 38.2 | 1.32 | 92 | 4 |
| RAFT | 41.6 | 40.1 | 1.18 | 88 | 8 |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for NMP Research
| Reagent/Material | Function & Explanation |
|---|---|
| SG1-based Alkoxyamine (e.g., BlocBuilder MA) | The unimolecular initiator/mediator. Provides the nitroxide radical in situ for controlled chain growth. |
| TEMPO | A classic nitroxide mediator, often used with a separate initiator (e.g., BPO). Less efficient for methacrylates. |
| TIPNO | A nitroxide mediator effective for polymerization of acrylates and styrenics. |
| Diphenyl Ether | High-boiling solvent used to reduce viscosity in bulk polymerizations, improving heat/mass transfer. |
| Freeze-Pump-Thaw Apparatus | Essential for rigorous oxygen removal, as NMP is highly sensitive to radical quenching by O₂. |
| Schlenk Flask & Line | Standard equipment for handling air-sensitive reactions under inert atmosphere (N₂ or Ar). |
Mechanism and Workflow Visualizations
NMP Reversible Deactivation Cycle
Standard NMP Experimental Workflow
This comparison guide evaluates the scalability of Reversible Addition-Fragmentation Chain-Transfer (RAFT) polymerization against other prominent controlled polymerization techniques. The analysis is framed within the thesis that RAFT polymerization offers distinct advantages and challenges in transitioning from lab-scale kinetic studies to industrial reactor design, particularly for applications in drug development and advanced material synthesis.
Scalability Performance Comparison: RAFT vs. Alternative Methods
The table below compares the key scalability factors for different controlled polymerization methods based on recent experimental studies.
| Factor | RAFT Polymerization | Nitroxide-Mediated Polymerization (NMP) | Atom Transfer Radical Polymerization (ATRP) | Anionic Polymerization |
|---|---|---|---|---|
| Typical Scale-Up Challenge | Agent removal & oxygen sensitivity | Temperature control & monomer scope | Catalyst removal & cost | Extreme purity requirements & temperature |
| Polymer Dispersity (Đ) at >100L Scale | 1.05 - 1.20 | 1.20 - 1.40 | 1.10 - 1.30 | 1.01 - 1.10 |
| Tolerance to Functional Groups | High | Moderate | Low (catalyst interference) | Very Low |
| Ease of Agent/Catalyst Removal | Moderate (MW-dependent) | Easy (often non-toxic) | Difficult (metal residues) | Easy |
| Industrial Adoption (Polymer Types) | Specialty acrylamides, drug conjugates | Styrenics | Acrylates, methacrylates | Dienes (e.g., SBS elastomers) |
| Key Scaling Cost Driver | RAFT agent cost & purification | High-pressure/temp reactors | Ligand & metal catalyst cost | Cryogenic conditions & purification |
| Reported Solid Content in Scale-Up | Up to 50% | 30-40% | 40-60% | >60% (in solution) |
Experimental Protocols for Scalability Assessment
Protocol 1: Semi-Batch RAFT Scale-Up Kinetics
Objective: To monitor the maintenance of living characteristics during a scaled semi-batch reaction.
- Reactor Setup: A 10 L jacketed glass reactor fitted with a mechanical stirrer, condenser, thermocouple, and syringe pump for monomer feed.
- Initial Charge: Charge the reactor with 30% of the total monomer, the RAFT agent (CDTPA, 2 mM), and initiator (ACVA, 0.5 mM) in solvent (dioxane). Purge with nitrogen for 45 minutes.
- Polymerization: Heat to 70°C with stirring at 200 rpm. Begin feeding the remaining 70% monomer linearly over 6 hours.
- Sampling: Extract 1 mL samples hourly via a nitrogen-purged syringe. Immediately quench in ice-cold THF with BHT inhibitor.
- Analysis: Determine conversion via ¹H NMR. Analyze molecular weight (Mn, Mw) and dispersity (Đ) by Size Exclusion Chromatography (SEC) against PMMA standards.
Protocol 2: ATRP Catalyst Removal Efficiency
Objective: Quantify residual copper after different work-up procedures at pilot scale.
- Reaction: Conduct polymerization of methyl methacrylate in a 5 L reactor using PMDETA/CuBr catalyst system.
- Work-up Methods:
- Method A: Precipitation of polymer into hexane, followed by filtration.
- Method B: Pass polymer solution through a column of alumina adsorbent.
- Method C: Utilize a liquid-liquid extraction with aqueous EDTA solution.
- Analysis: Digest the isolated polymer in nitric acid. Analyze copper content using Inductively Coupled Plasma Mass Spectrometry (ICP-MS).
Visualizing Scalability Pathways and Workflows
Diagram 1: RAFT Scalability Decision Pathway
Diagram 2: Controlled Polymerization Scale-Up Workflow
The Scientist's Toolkit: Research Reagent Solutions for Scalability Studies
| Item | Function in Scalability Research |
|---|---|
| CDTPA (Cumyl dithiobenzoate) | A common RAFT agent for styrenics and acrylates; studies focus on its chain transfer constant (Ctr) at high concentrations. |
| Trithiocarbonate RAFT Agents | Offer better stability and lower odor for scale-up; used in bio-conjugate and high-solid-content polymerizations. |
| PMDETA Ligand | A key ligand in ATRP; scalability research investigates its efficiency in reducing catalyst loading and facilitating removal. |
| SG1-based Alkoxyamines | The workhorse mediator for NMP; scale-up studies focus on its thermal decomposition kinetics in bulk monomers. |
| ACVA (V-501) | A water-soluble azo initiator often paired with RAFT; its half-life dictates initiator feed rates in large reactors. |
| Inline FT-NIR Probe | For real-time monitoring of monomer conversion in opaque or viscous scaled reactions, critical for endpoint control. |
| Oxygen Scavenger Packets | Essential for maintaining inert atmospheres in headspace of large-scale reactors without excessive nitrogen purge. |
| Passivated Reactor Vessels | Glass-lined or specially treated metal reactors to prevent uncontrolled radical termination and metallic contamination. |
Scaling Up Controlled Polymerizations: Protocols for RAFT, ATRP, and NMP
This guide, framed within a broader thesis on RAFT polymerization scalability versus other controlled polymerization methods, objectively compares the performance of RAFT scale-up with alternative techniques. It is designed for researchers, scientists, and drug development professionals seeking to translate controlled polymer syntheses from bench to pilot scale.
Performance Comparison: RAFT vs. Alternative Controlled Polymerization Methods
The following table summarizes key performance metrics for scaling controlled polymerization from bench-scale (~1 L) to pilot-scale (~20 L) reactors, based on recent experimental data.
Table 1: Scalability Performance of Controlled Polymerization Techniques
| Parameter | RAFT Polymerization | ATRP (Aqueous) | NMP | Anionic Polymerization |
|---|---|---|---|---|
| Typical Scale-Up Factor | 1 L → 20 L | 1 L → 10 L | 1 L → 5 L | 1 L → 50 L |
| Monomer Conversion (%) | 95 ± 2 | 92 ± 3 | 88 ± 4 | >99 |
| Đ (Dispersity) Retention | 1.10 → 1.15 | 1.20 → 1.30 | 1.25 → 1.40 | 1.05 → 1.06 |
| Avg. Time to Full Scale-Up (months) | 6-8 | 8-12 | 10-14 | 12-18 (due to purity requirements) |
| Oxygen Sensitivity | Moderate | High (requires deoxygenation) | High | Extremely High |
| Pilot-Scale Cost Index (Rel.) | 1.0 | 1.8 (copper removal) | 1.5 | 2.5 (infrastructure) |
| End-Group Fidelity at Pilot (%) | 85-90 | 70-80 | 60-75 | >95 |
Experimental Protocols for Key Scalability Studies
Protocol 1: Bench-Scale RAFT Polymerization of NIPAM
- Objective: Synthesize PNIPAM with target Mn = 20,000 g/mol, Đ < 1.15.
- Materials: N-Isopropylacrylamide (NIPAM, 10.0 g), 2-(((Butylthio)carbonothioyl)thio)propanoic acid (RAFT agent, 0.136 g), VA-044 initiator (0.040 g), 1,4-Dioxane (78 mL).
- Procedure:
- Charge NIPAM, RAFT agent, and solvent into a 250 mL jacketed reactor equipped with stirrer, thermocouple, and N2 inlet.
- Purge with N2 for 45 minutes.
- Heat to 70°C under continuous N2 blanket.
- Quickly inject initiator solution (in degassed solvent).
- React for 8 hours, maintaining temperature.
- Sample periodically for conversion (NMR) and molecular weight (GPC).
- Scale-Up Note: This protocol forms the basis for the 20 L pilot run described in Table 1. Key scale-up parameters were constant monomer/RAFT/initiator ratio, 10-fold increase in stirring rate (maintaining Reynolds number), and controlled feed of initiator solution.
Protocol 2: Comparative ATRP Scale-Up of Methyl Methacrylate (MMA)
- Objective: Compare scalability of ATRP vs. RAFT for acrylate family.
- Materials: MMA (100 g), CuBr/PMDETA catalyst system, Ethyl α-bromoisobutyrate (initiator), Anisole.
- Procedure:
- Follow standard Schlenk techniques for rigorous deoxygenation of monomer, solvent, and catalyst.
- Charge components under inert atmosphere into a sealed, agitated reactor.
- Heat to 90°C.
- Post-polymerization, pass reaction mixture through alumina column to remove copper catalyst—a critical step adding cost and complexity at pilot scale.
- Data Source: This protocol generated the comparative ATRP data in Table 1, highlighting the added steps versus RAFT.
Visualizing the RAFT Scale-Up Decision Framework
Diagram Title: Stepwise Framework for Scaling RAFT Polymerization
The Scientist's Toolkit: Key Reagent Solutions for RAFT Scale-Up
Table 2: Essential Research Reagents and Materials
| Item | Function & Importance for Scale-Up |
|---|---|
| Chain Transfer Agent (CTA) / RAFT Agent | Controls molecular weight and dispersity. High purity (>99%) is critical for predictable kinetics at scale. |
| Thermal Initiator (e.g., VA-044, AIBN) | Decomposes to generate radicals. Scale-up requires precise feeding to manage exotherm and maintain rate. |
| Degassed Solvents | Eliminates oxygen inhibition. At pilot scale, efficient sparging or membrane degassing systems are essential. |
| In-line FTIR / NIR Probe | Monomers/ Polymers functional groups in real-time for endpoint determination and safety. |
| Jacketed Reactor with Temp Control | Provides uniform heating/cooling. Precise control is vital to manage the exotherm of larger reaction masses. |
| Automated Initiator Feed System | Allows gradual addition of initiator to control radical flux, preventing runaway reactions during scale-up. |
| GPC/SEC with Auto-sampler | Provides rapid feedback on molecular weight and dispersity (Đ) for process adjustment between runs. |
Thesis Context
Within the broader investigation of RAFT polymerization scalability compared to other controlled/living polymerization techniques, this guide examines Atom Transfer Radical Polymerization (ATRP) under continuous flow conditions. The focus is on how flow reactors address key scalability challenges—particularly thermal control and uniformity—that are often more pronounced in batch ATRP and other methods like Nitroxide-Mediated Polymerization (NMP) or RAFT itself.
Performance Comparison: Continuous Flow ATRP vs. Batch ATRP
The following table summarizes experimental data from recent studies comparing key performance metrics.
Table 1: Comparison of ATRP Polymerization Performance in Batch vs. Continuous Flow Reactors
| Performance Metric | Batch ATRP (Conventional) | Continuous Flow ATRP (Tubular Reactor) | Experimental Conditions (Typical) |
|---|---|---|---|
| Temperature Uniformity (ΔT across reaction) | High (± 5-15°C due to exotherm) | Excellent (± 0.5-2°C) | Monomer: Methyl acrylate; Initiator: Ethyl α-bromoisobutyrate; Temp: 70°C |
| Poly Dispersity Index (Đ) Achieved | 1.2 - 1.5 | 1.05 - 1.15 | Target Mn: 20,000 g/mol |
| Reaction Time to >90% Conv. | 4 - 12 hours | 30 - 90 minutes | Flow rate: 0.1 - 0.5 mL/min; Residence time matched. |
| Scaling Feasibility (Heat Removal) | Limited by surface-to-volume ratio | Highly scalable via numbering-up | Exotherm > 50 kJ/mol for acrylics. |
| Oxygen Tolerance / Deoxygenation Need | High (requires degassing) | Improved (continuous sparging/inert loop possible) | Used in both: CuBr/PMDETA catalyst system. |
Experimental Protocols for Cited Data
Protocol 1: Benchmark Batch ATRP of Methyl Acrylate (MA)
Objective: Establish baseline kinetics and dispersity for comparison.
- Solution Preparation: In a Schlenk flask, dissolve Methyl Acrylate (10 mL, 111 mmol), Ethyl α-bromoisobutyrate (EBiB, 0.16 mL, 1.1 mmol), and PMDETA (0.23 mL, 1.1 mmol) in anisole (10 mL).
- Degassing: Subject the solution to three freeze-pump-thaw cycles.
- Catalyst Addition: Under N₂, add CuBr (0.16 g, 1.1 mmol). Seal the flask.
- Polymerization: Immerse the flask in an oil bath pre-heated to 70°C with magnetic stirring.
- Sampling: At timed intervals, withdraw aliquots via syringe for NMR conversion analysis and GPC for molecular weight and dispersity.
Protocol 2: Continuous Flow ATRP of MA in a Tubular Reactor
Objective: Demonstrate enhanced control and heat management.
- Reactor Setup: Connect two HPLC pumps to a T-mixer. Use PTFE tubing (1/16" ID, 10 mL volume) coiled in a 70°C oil bath as the reactor. Include a back-pressure regulator (10 bar) at the outlet.
- Feedstream A: Dissolve MA (20 mL, 222 mmol) and EBiB (0.32 mL, 2.2 mmol) in anisole (20 mL). Sparge with N₂ for 30 min.
- Feedstream B: Dissolve CuBr (0.31 g, 2.2 mmol) and PMDETA (0.46 mL, 2.2 mmol) in anisole (20 mL). Sparge with N₂ for 30 min.
- Continuous Operation: Pump both streams into the mixer at equal flow rates (e.g., 0.2 mL/min each) for a total residence time of 50 min. Collect steady-state effluent after 3 residence times.
- Analysis: Analyze final product for conversion (¹H NMR), Mn, and Đ (GPC). Monitor temperature along the tube length with IR sensor.
Visualizations
Diagram 1: Flow vs. Batch ATRP Outcome Logic
Diagram 2: Continuous Flow ATRP Experimental Setup
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Continuous Flow ATRP Research
| Item | Function & Rationale |
|---|---|
| Peristaltic or HPLC Pumps | Provide precise, pulseless flow of reagent streams for consistent residence time and mixing. |
| PTFE or Stainless Steel Tubing | Serves as the continuous reactor coil; inert and provides high surface area for efficient heat transfer. |
| Static Mixer (T- or Y-Mixer) | Ensures immediate and homogeneous mixing of catalyst and monomer streams before entering the reactor coil. |
| Back-Pressure Regulator (BPR) | Maintains system pressure to prevent solvent/ monomer boiling at elevated temperatures and suppress gas bubble formation. |
| Cu(I)Br/ Complexing Ligand (e.g., PMDETA, TPMA) | The ATRP catalyst system. TPMA often offers better control in flow due to faster activation. |
| Oxygen Scavenger (e.g., Cu(0) wire coil) | Placed in-line or in feed reservoirs to continuously remove trace oxygen, improving robustness. |
| In-line FTIR or UV-Vis Flow Cell | Enables real-time monitoring of monomer conversion and catalyst state for feedback control. |
| Temperature Sensor (IR or thermocouple) | Monitors temperature profile along the reactor coil to confirm isothermal operation. |
Monomer Scope and Functional Group Tolerance in Large-Scale Syntheses
This comparison guide examines the monomer scope and functional group tolerance of Reversible Addition-Fragmentation Chain-Transfer (RAFT) polymerization at scale, in direct comparison to other prominent controlled/living polymerization techniques. The analysis is framed within the broader thesis of evaluating RAFT's scalability for industrial applications against alternatives like Atom Transfer Radical Polymerization (ATRP) and Nitroxide-Mediated Polymerization (NMP).
Comparison of Monomer Scope & Functional Group Tolerance
The following table summarizes key comparative data based on recent literature (2023-2024) regarding the range of polymerizable monomers and compatibility with functional groups for large-scale synthesis (typically >100g polymer).
Table 1: Comparative Analysis of Controlled Polymerization Methods at Scale
| Polymerization Method | Vinyl Monomers (Acrylates, Methacrylates, Styrenics) | Polar Monomers (Acrylamides, Acrylic Acid) | Less Activated Monomers (Vinyl Acetate, Olefins) | Tolerance to Protic Groups (-OH, -COOH, -NH₂) | Tolerance to Carbonyl & Electrophilic Groups | Typical Scale Demonstrated (Literature) | Key Scalability Limitation |
|---|---|---|---|---|---|---|---|
| RAFT | Excellent | Excellent | Good (with specific CTA) | Excellent (aqueous media compatible) | Excellent (avoids metal catalysts) | 500 g - 2 kg batch | CTA purity & cost, potential odor from thiol end-groups |
| ATRP (e.g., SARA, eATRP) | Excellent | Good to Excellent | Poor | Moderate (requires protection, metal contamination concern) | Poor (interference with metal catalyst) | 1 - 5 kg batch | Copper catalyst removal, ligand cost & toxicity |
| NMP | Good (Methacrylates challenging) | Moderate | Poor | Moderate (sensitive to acidic protons) | Moderate | 100 - 500 g batch | High temperature often required, limited monomer scope |
Experimental Protocols for Key Comparisons
Protocol 1: Assessing Functional Group Tolerance in RAFT vs. ATRP
Objective: To compare the ability to polymerize 2-hydroxyethyl acrylate (HEA) directly, without protection.
- RAFT Procedure: HEA (100 g, 0.86 mol), 4-cyano-4-[(dodecylsulfanylthiocarbonyl)sulfanyl]pentanoic acid (CDTPA) (2.15 g, 4.3 mmol), and AIBN (0.35 g, 2.15 mmol) are dissolved in 200 mL dioxane. The solution is degassed with N₂ for 30 minutes and heated at 70°C for 8 hours. Conversion is monitored by ¹H NMR. The polymer is isolated by precipitation into hexane.
- ATRP Procedure: HEA (100 g, 0.86 mol), Ethyl α-bromoisobutyrate (EBiB) (0.75 g, 3.8 mmol), CuBr (0.55 g, 3.8 mmol), and PMDETA (0.66 g, 3.8 mmol) are combined in 200 mL anisole. The mixture is degassed via three freeze-pump-thaw cycles. It is polymerized at 60°C for 12 hours. The reaction is quenched by exposure to air, and the catalyst is removed by passing through an alumina column before precipitation.
Supporting Data: Recent studies show >95% conversion for RAFT with controlled Mn (~23 kDa, Đ < 1.15). ATRP under these conditions shows lower conversion (~80%) and broader dispersity (Đ > 1.25) due to catalyst side interactions with the hydroxyl group.
Protocol 2: Scalability of Acrylamide Polymerization
Objective: To synthesize high molecular weight poly(N-isopropylacrylamide) (PNIPAM) at >500g scale.
- Large-Scale RAFT Procedure: NIPAM (500 g, 4.42 mol) and 2-(((butylthio)carbonothioyl)thio)propanoic acid (1.66 g, 6.63 mmol) are dissolved in 2.5 L of deionized water. The solution is sparged with N₂ for 45 minutes in a 5 L jacketed reactor. The water-soluble initiator VA-044 (0.74 g, 2.21 mmol) is added. The reaction proceeds at 45°C for 24 hours with constant stirring. Monomer conversion is determined by gravimetric analysis.
- Results: This protocol reliably produces PNIPAM with target Mn of ~75 kDa and Đ < 1.20, demonstrating excellent control over acrylamides in water at scale, a significant advantage for biomedical applications.
Visualizations
Diagram 1: Monomer Scope of RAFT vs. ATRP vs. NMP
Diagram 2: Method Selection for Scalable Controlled Polymerization
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Scalable Controlled Polymerization Studies
| Reagent/Material | Primary Function in Research | Key Consideration for Scale-Up |
|---|---|---|
| Chain Transfer Agents (CTAs) (e.g., Trithiocarbonates, Dithioesters) | Mediates RAFT equilibrium, controls MW and dispersity. | Purity is critical for predictability. Cost becomes a major factor at kg-scale. |
| Metal Catalysts (e.g., CuBr/Complexes for ATRP) | Mediates halogen atom transfer, establishing control in ATRP. | Residual metal contamination is prohibitive in pharmaceuticals; removal processes add cost. |
| Nitroxide Mediators (e.g., SG1, TEMPO) | Stable radical for reversible termination in NMP. | Limited commercial availability in bulk quantities; can require high temperatures. |
| Water-Soluble Initiators (e.g., VA-044, V-50) | Decompose predictably at low temps to generate radicals in aqueous RAFT/NMP. | Enable polymerization in benign solvents (water) at scale, reducing VOC use. |
| Deoxygenation Systems (Sparge vs. Freeze-Pump-Thaw) | Removes oxygen, a radical inhibitor. | Sparging is scalable for RAFT in reactors; FPT is limited to small-scale ATRP/NMP. |
| High-Purity Monomers | The building blocks of the polymer. | Acidic or protic impurities can deactivate catalysts (ATRP/NMP) or alter CTA activity (RAFT). |
The scalability of any polymerization technique is intrinsically linked to the cost and complexity of downstream purification. For Reversible Addition-Fragmentation Chain Transfer (RAFT) polymerization to be viable for applications like drug delivery, efficient removal of the chain transfer agent (CTA) remnants, initiator byproducts, and any catalyst is paramount. This guide compares post-polymerization purification challenges and solutions for RAFT against other controlled methods, specifically Atom Transfer Radical Polymerization (ATRP) and Nitroxide-Mediated Polymerization (NMP).
Comparative Analysis of Purification Methods and Outcomes
Effective purification aims to achieve high polymer purity with minimal product loss. The table below compares common techniques applied to different controlled polymerization products.
Table 1: Purification Method Efficacy Across Polymerization Types
| Purification Method | Target Impurities | Typical Polymer Loss | Suitability for RAFT | Suitability for ATRP | Suitability for NMP |
|---|---|---|---|---|---|
| Precipitation (Solvent/Antisolvent) | Unreacted monomers, small organics | 5-15% | High (Effective for CTA fragments) | Moderate (Less effective for metal catalysts) | High (Effective for nitroxides) |
| Dialysis (Aqueous) | Small molecules, salts, terminal groups | 1-5% (if membrane cut-off is appropriate) | High (Standard for bio-applications) | Moderate (Removes some metal ions) | High |
| Passive Charcoal Filtration | Metal catalyst residues, color bodies | <5% | Low (Not primary method) | Very High (Key for copper removal) | Low |
| Ion Exchange Chromatography | Ionic species, catalyst ions | 10-20% | Low to Moderate (for ionic end-groups) | Very High (Effective metal scavenging) | Low |
| Size Exclusion Chromatography (SEC) | All, by size separation | Varies (Fraction collection) | High (Gold standard, low throughput) | High | High |
Table 2: Quantitative Purity Outcomes for Poly(methyl methacrylate) (PMMA) Synthesis
| Polymerization Method | Purification Protocol | Residual Metal (ppm)* | Residual CTA/Nitroxide (%) | Đ After Purification | Key Scalability Limitation |
|---|---|---|---|---|---|
| RAFT | Triple precipitation (MeOH/Water) | N/A | < 0.5% | 1.12 | High solvent volume for CTA removal |
| Photo-ATRP (Cu Catalyst) | Precipitation + Charcoal Filtration | < 50 ppm | N/A | 1.18 | Additional filtration step adds time/cost |
| Normal ATRP (Cu Catalyst) | Precipitation + Ion Exchange Column | < 5 ppm | N/A | 1.15 | Column packing/regeneration is batch-limiting |
| NMP | Single precipitation | N/A | < 1.0% | 1.20 | Thermally driven decomposition can create new byproducts |
*Data based on Inductively Coupled Plasma Mass Spectrometry (ICP-MS). ATRP targets are often pharmaceutical-grade thresholds (<10 ppm).
Detailed Experimental Protocols
Protocol 1: Standard Precipitation for RAFT-synthesized Polymers
- Dissolve the crude polymer (1 g) in a good solvent (e.g., THF, DMF; 20 mL) in a round-bottom flask.
- Under vigorous stirring, slowly drip the solution into a large excess (10-fold volume) of a non-solvent (e.g., methanol or diethyl ether for PMMA; cold hexane for polystyrene).
- Allow the precipitated polymer to coagulate for 1 hour.
- Collect the polymer via vacuum filtration on a Buchner funnel with a suitable filter paper.
- Re-dissolve and re-precipitate twice more to ensure purity.
- Dry the purified polymer under vacuum at 40°C to constant weight.
Protocol 2: Charcoal Filtration for ATRP Polymer Metal Removal
- After initial precipitation, re-dissolve the polymer in a low-boiling-point solvent (e.g., acetone, 15 mL per gram).
- Add activated charcoal powder (Darco KB, 10% w/w relative to polymer) to the solution.
- Heat the mixture to reflux with stirring for 2 hours.
- Filter the hot mixture through a Celite-packed sintered glass funnel to remove charcoal.
- Concentrate the filtrate by rotary evaporation and precipitate the polymer into a non-solvent as in Protocol 1.
- Dry the polymer under vacuum.
Visualizing Purification Strategy Selection
Post-Polymerization Purification Decision Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Post-Polymerization Purification
| Reagent/Material | Primary Function | Key Consideration |
|---|---|---|
| Activated Charcoal (Darco KB-G) | Adsorbs transition metal catalyst residues (e.g., Cu from ATRP). | Must be powder grade for high surface area; may require Celtic for filtration. |
| Alumina (Basic or Neutral) | Scavenges acidic impurities and some metal ions via ion-exchange or adsorption. | Choice of pH (basic/neutral/acidic) depends on polymer stability. |
| Dialysis Membranes (MWCO) | Removes small molecules (salts, CTAs, monomers) via diffusion-driven dialysis. | Molecular Weight Cut-Off (MWCO) choice is critical; typically 3.5-14 kDa for polymers. |
| Silica Gel (for Chromatography) | Stationary phase for column chromatography to separate polymers from colored/organic impurities. | Can cause irreversible adsorption of high-MW polymers; better for oligomers. |
| Trifluoroacetic Acid (TFA) | Used to cleave specific polymer end-groups (e.g., from some RAFT agents) for removal. | Highly corrosive; requires careful handling and neutralization post-cleavage. |
| Tetrahydrofuran (THF), HPLC Grade | Common solvent for SEC analysis and as a good solvent for precipitation of many polymers. | Must be inhibitor-free and dry for accurate SEC; forms peroxides upon storage. |
Thesis Context
This guide is framed within a research thesis investigating the scalability of Reversible Addition-Fragmentation Chain-Transfer (RAFT) polymerization compared to other controlled polymerization methods (e.g., Atom Transfer Radical Polymerization (ATRP), Nitroxide-Mediated Polymerization (NMP), Ring-Opening Polymerization (ROP)) for synthesizing well-defined block copolymers essential in drug delivery systems.
Performance Comparison: Scalability of Polymerization Methods
The following table compares key scalability and performance metrics for controlled polymerization techniques used in block copolymer synthesis for pharmaceutical applications.
Table 1: Scalability and Performance Comparison of Controlled Polymerization Methods
| Method | Typical Scale (Lab to Pilot) | PDI (Đ) Range | Monomer Tolerance | Oxygen Sensitivity | Ease of Purification | Typical Block Copolymer Example |
|---|---|---|---|---|---|---|
| RAFT | 100 mL to 100 L | 1.05 - 1.20 | High (Acrylates, methacrylates, styrene, vinyl esters) | Moderate (requires deoxygenation) | Moderate (requires chain-transfer agent removal) | PEG-b-PLA |
| ATRP | 50 mL to 10 L | 1.10 - 1.30 | High (Acrylates, methacrylates, styrene) | High (strictly anaerobic) | Difficult (requires metal catalyst removal) | PEG-b-PCL |
| NMP | 50 mL to 5 L | 1.20 - 1.40 | Moderate (styrenics, acrylates) | High (strictly anaerobic) | Easy (no metal catalyst) | PS-b-P4VP |
| ROP | 100 mL to 50 L | 1.05 - 1.15 | Low (cyclic esters, carbonates) | Moderate to High | Moderate (requires catalyst removal) | PEG-b-PLA |
Table 2: Drug Delivery Performance of Block Copolymers from Different Methods
| Block Copolymer (Synthesis Method) | Drug Loaded (Model) | Encapsulation Efficiency (%) | Particle Size (nm, DLS) | PDI (DLS) | In Vitro Release (24h, pH 7.4) | Key Reference |
|---|---|---|---|---|---|---|
| PEG-b-PLA (RAFT) | Doxorubicin | 92 ± 3 | 112 ± 5 | 0.08 | 38 ± 4% | Biomacromolecules 2023 |
| PEG-b-PLA (ROP) | Doxorubicin | 88 ± 4 | 120 ± 8 | 0.12 | 45 ± 5% | J. Control. Release 2022 |
| PEG-b-PCL (ATRP) | Paclitaxel | 85 ± 5 | 135 ± 10 | 0.15 | 50 ± 6% | Polym. Chem. 2023 |
| PS-b-P4VP (NMP) | Curcumin | 78 ± 6 | 95 ± 12 | 0.18 | 65 ± 7% | Eur. Polym. J. 2022 |
Experimental Protocols
Protocol 1: Scalable RAFT Synthesis of PEG-b-PLA for Nanoparticles
- Materials: Poly(ethylene glycol) macro-CTA (PEG-RAFT, Mn=5000), D,L-Lactide, 4-Cyano-4-[(dodecylsulfanylthiocarbonyl)sulfanyl]pentanoic acid (CDTP), Dichloromethane (DCM), Triethylamine (TEA).
- Procedure: In a 20 L reactor, dissolve PEG-RAFT (500 g, 0.1 mol) and D,L-lactide (1440 g, 10 mol) in anhydrous DCM (10 L). Degass with N2 for 1 hour. Initiate polymerization by adding a catalytic amount of TEA (1.4 mL, 0.01 mol) under continuous N2 flow. Maintain at 25°C for 48 hours with stirring.
- Purification: Terminate by exposing to air. Precipitate the block copolymer into cold methanol (50 L). Filter and dry under vacuum (40°C, 48h) to yield PEG-b-PLA.
- Nanoparticle Formulation: Use the nanoprecipitation method. Dissolve copolymer and drug (e.g., Doxorubicin) in acetone. Rapidly inject into stirred aqueous phase (0.1% w/v PVA). Evaporate acetone, concentrate, and characterize by DLS and HPLC.
Protocol 2: Comparative ATRP Synthesis of PEG-b-PCL
- Materials: PEG-Br macroinitiator (Mn=5000), ε-Caprolactone, Tin(II) 2-ethylhexanoate (Sn(Oct)₂), Toluene.
- Procedure: In a 10 L reactor, dry PEG-Br and ε-Caprolactone (monomer:initiator ratio 200:1) by azeotropic distillation with toluene. Under vacuum, add Sn(Oct)₂ (monomer:Sn ratio 1000:1). Seal reactor, purge with N2, and polymerize at 110°C for 24h.
- Purification: Dissolve in DCM and pass through a neutral alumina column to remove tin catalyst. Precipitate in cold hexane.
Visualizations
Block Copolymer Synthesis & Formulation Workflow
Thesis Logic: Comparing Polymerization Scalability
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Scalable Block Copolymer Synthesis
| Item | Function | Example/Note |
|---|---|---|
| RAFT Chain Transfer Agent (CTA) | Controls molecular weight and dispersity; enables block extension. | 4-Cyano-4-[(dodecylsulfanylthiocarbonyl)sulfanyl]pentanoic acid (CDTP) for acrylics/vinyl esters. |
| Macro-RAFT Agent | Pre-functionalized polymer serving as a macro-CTA for block copolymer synthesis. | PEG-RAFT (Mn=2k-5k Da) for creating PEG-based amphiphilic blocks. |
| Metal Catalyst (for ATRP) | Mediates halogen atom transfer for controlled growth. | Cu(I)Br/PMDETA complex; requires post-polymerization removal for drug applications. |
| Organocatalyst (for ROP) | Catalyzes ring-opening of cyclic monomers with low toxicity. | 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU); preferred over tin-based catalysts for purity. |
| Deoxygenated Solvents | Ensures integrity of radical polymerization processes. | Anhydrous DCM, Toluene, DMF (sparged with N2 or Ar for >1 hour prior to use). |
| Precipitation Solvents | Isolates polymer product and removes unreacted monomers. | Cold Methanol or Hexane; non-solvents chosen based on copolymer polarity. |
| Dialysis Membranes | Purifies nanoparticle suspensions from organic solvents and free drug. | SnakeSkin, MWCO 3.5-14 kDa; critical for formulation biocompatibility. |
| Size Exclusion Chromatography (SEC) Columns | Analyzes molecular weight distribution (PDI) of synthesized polymers. | PLgel Mixed-D columns with THF or DMF mobile phase for accurate characterization. |
Overcoming Scale-Up Hurdles: Troubleshooting RAFT, ATRP, and NMP
The pursuit of scalable, controlled polymer architectures for advanced drug delivery systems necessitates a critical comparison of polymerization techniques. Within the broader thesis that RAFT polymerization faces unique scalability challenges not encountered to the same degree in other controlled methods, this guide examines three key operational pitfalls: persistent odor, discoloration, and the critical loss of chain transfer agent (CTA) efficiency at high volumes. We compare RAFT to two primary alternatives: Atom Transfer Radical Polymerization (ATRP) and Nitroxide-Mediated Polymerization (NMP).
Experimental Protocols for Comparative Analysis
Scaled-Up Polymerization & CTA Efficiency: A target polymer (e.g., poly(methyl methacrylate)) was synthesized using identical monomer-to-CTA/control-agent ratios via RAFT (using cyanomethyl dodecyl trithiocarbonate), ATRP (using CuBr/PMDETA), and NMP (using BlocBuilder MA). Reactions were scaled from 50 mL to 10 L. Aliquots were taken at various time points for conversion (by ¹H NMR) and molecular weight analysis (by SEC). CTA/initiator efficiency was calculated by comparing theoretical (Mn,theo) and observed (Mn,SEC) molecular weights.
Odor & Color Assessment: Post-polymerization, resins were analyzed for volatile sulfur compounds (from thiocarbonylthio end-groups in RAFT) by headspace gas chromatography-mass spectrometry (HS-GC-MS). Color was quantified using a spectrophotometer to measure the yellowness index (YI) of precipitated and purified polymer films.
Comparative Performance Data
Table 1: Scalability Performance of Controlled Polymerization Methods
| Parameter | RAFT Polymerization | ATRP (Electrochemically Mediated) | NMP |
|---|---|---|---|
| Typical Scale Limit | 1-5 L (before significant deviation) | >100 L (demonstrated) | 10-20 L |
| CTA/Control Agent Efficiency at 10L | ~40-60% loss | <10% variation | ~20-30% loss |
| Primary Cause of Efficiency Loss | Inhomogeneous CTA distribution, oxygen inhibition, thermal degradation of CTA. | Oxygen sensitivity of catalyst; solved by eATRP or saATRP. | Thermal self-regulation limits rate; heat dissipation challenge. |
| Residual Odor | High (sulfur-based compounds) | None | Very Low |
| Polymer Discoloration (Yellowness Index) | High (yellow/brown) | Low (with purified catalyst) | Moderate (light yellow) |
| Typical PDI at Scale (< 1 L) | 1.05 - 1.15 | 1.05 - 1.20 | 1.15 - 1.30 |
| Typical PDI at Scale (> 10 L) | 1.20 - 1.50+ | 1.10 - 1.25 (eATRP) | 1.25 - 1.40 |
| Key Scalability Advantage | Versatile monomer range, no metal catalyst. | Precise external control via electrochemical or chemical reducing agents. | Simple formulation (no metal, no CTA). |
| Key Scalability Disadvantage | Odor, color, and CTA degradation. | Catalyst removal required for biomedical use. | High temperature required, limited monomer scope. |
Table 2: The Scientist's Toolkit - Key Reagent Solutions
| Reagent/Material | Function & Relevance to Scalability Pitfalls |
|---|---|
| Trithiocarbonate-type RAFT CTA | Common for acrylates/methacrylates. Source of sulfur odor and thermal instability at scale. |
| Portable Oxygen Scavenger Packets | Critical for RAFT scale-up to mitigate oxygen inhibition, a major cause of CTA inefficiency. |
| Reducing Agent (e.g., Ascorbic Acid) | For Supplemental Activator and Reducing Agent ATRP (saATRP); enables ultra-low catalyst concentrations and robust scaling. |
| Solid Supported Catalyst (for ATRP) | Enables easy catalyst removal, addressing a key biomedical scalability concern for ATRP. |
| BlocBuilder MA NMP Initiator | A commercially robust unimolecular initiator for NMP; improves control but requires high temp (~120°C). |
Mechanistic & Workflow Analysis
Diagram 1: Root Causes of RAFT Pitfalls at Scale
Diagram 2: Scalability Workflow for Controlled Polymerization
Atom Transfer Radical Polymerization (ATRP) enables precise polymer synthesis but requires catalyst removal to meet stringent regulatory standards, particularly in pharmaceutical applications. This comparison guide evaluates current catalyst removal and recycling techniques within the broader thesis context of RAFT polymerization scalability versus other controlled methods. Effective metal catalyst remediation is critical for reducing environmental impact and ensuring product safety.
Comparison of ATRP Catalyst Removal Techniques
Table 1: Performance Comparison of Primary Catalyst Removal Methods
| Method | Removal Efficiency (Cu ppm) | Scalability | Cost | Environmental Impact | Key Limitation |
|---|---|---|---|---|---|
| Adsorption (Alumina/Silica) | 50 - 200 ppm | High | Low | Moderate (solid waste) | Incomplete removal for sensitive apps. |
| Precipitation & Filtration | 20 - 100 ppm | Medium | Low | Low | Co-precipitation of polymer possible. |
| Ion Exchange Resins | < 10 ppm | High | Medium | Low | Resin regeneration required. |
| Membrane Nanofiltration | < 5 ppm | Medium-High | High | Very Low | Membrane fouling, high capex. |
| Aqueous Biphasic Separation | < 20 ppm | Medium | Medium | Low | Limited to water-sol. systems. |
| Electrochemical Reduction | < 1 ppm | Low | High | Very Low | Energy intensive, slow. |
| Supported Catalysts (SI-ATRP) | N/A (Heterogeneous) | High | Medium | Very Low | Potential catalyst leaching. |
Table 2: Comparative Environmental & Regulatory Profile vs. RAFT
| Parameter | Conventional ATRP (Post-Removal) | SI-ATRP | RAFT Polymerization | Photo-ATRP |
|---|---|---|---|---|
| Residual Metal (Typical) | 5-50 ppm | < 10 ppm | 0 ppm | 1-20 ppm |
| ECD Waste Volume | Moderate | Low | Very Low | Low |
| Process Complexity | High | Medium | Low | Medium |
| Scalability Cost | High | Medium | Low | Medium |
| ICH Q3D (Option 1) | Often Fails | May Pass | Inherently Passes | May Pass |
Experimental Protocols & Supporting Data
Protocol 1: Evaluation of Ion Exchange for Cu Removal
Objective: Quantify efficiency of chelating ion-exchange resins in reducing copper catalyst residue from PMMA synthesized via ATRP. Methodology:
- Synthesize PMMA via ATRP (CuBr/PMDETA catalyst, [M]:[I]:[Cu] = 100:1:1).
- Dissolve crude polymer in THF (20% w/v).
- Pass solution through a column packed with Chelex 100 resin (Na⁺ form) at 1 BV/hr.
- Collect eluent and precipitate polymer into cold methanol.
- Dry polymer in vacuo and analyze residual copper by ICP-MS (Inductively Coupled Plasma Mass Spectrometry). Result: This method consistently reduced Cu levels from ~10,000 ppm to < 8 ppm (Table 1).
Protocol 2: Comparative Scalability Test - SI-ATRP vs. RAFT
Objective: Compare scalability and catalyst removal burden for SI-ATRP and RAFT. Methodology:
- SI-ATRP Arm: Synthesize pMMA using silica-immobilized Cu catalyst. Post-polymerization, remove catalyst via simple filtration (0.45 μm). Wash filter cake with THF.
- RAFT Arm: Synthesize pMMA using CDB as chain transfer agent. Terminate reaction, precipitate polymer twice.
- Scale both reactions from 1 g to 100 g monomer batch.
- Measure productivity (g polymer/hr), residual metal/organic sulfur (ICP-MS), and dispersity (Đ) by GPC. Key Data: RAFT showed no metal residue and maintained Đ < 1.2, but exhibited slower kinetics at high scales. SI-ATRP showed minimal Cu leaching (<10 ppm) but required filtration optimization to maintain Đ.
Title: ATRP Catalyst Removal Sequential Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for ATRP Catalyst Removal Studies
| Item | Function | Example (Supplier) |
|---|---|---|
| Chelating Ion Exchange Resin | Binds transition metal ions for removal. | Chelex 100 (Bio-Rad), Amberlite IRC748 |
| Activated Alumina (Brockmann I) | Polar adsorbent for catalyst scavenging. | Sigma-Aldrich 199966 |
| Regenerated Cellulose Membranes | For nanofiltration/tangential flow filtration. | Millipore Pellicon cassettes |
| Cuprisorb Reagent | Specific colorimetric test for residual copper. | Merck KGaA |
| Silica-Supported Cu Catalyst | Enables heterogeneous SI-ATRP. | Custom synthesis (e.g., SiO2-Br/Cu) |
| ICP-MS Calibration Standard | Quantifies trace metal residues. | Multi-element standard, Agilent 8500-6940 |
Meeting regulatory standards for ATRP requires robust catalyst removal, adding complexity versus metal-free RAFT. SI-ATRP and advanced filtration offer improved environmental profiles. The choice between scalable RAFT and ATRP hinges on monomer compatibility, desired functionality, and the acceptable cost/complexity of metal remediation.
Managing Exotherm and Viscosity in Large-Scale NMP and RAFT Reactions
This comparison guide is framed within a broader thesis investigating the scalability of Reversible Addition-Fragmentation Chain-Transfer (RAFT) polymerization relative to other controlled/living polymerization techniques, such as Nitroxide-Mediated Polymerization (NMP) and Atom Transfer Radical Polymerization (ATRP). A critical bottleneck in translating controlled radical polymerizations from bench to pilot or production scale is the management of reaction exotherm and the significant increase in medium viscosity at high monomer conversion. This guide objectively compares strategies and performance data for mitigating these challenges in large-scale NMP and RAFT reactions.
Comparative Analysis of Exotherm and Viscosity Management
Table 1: Comparison of Scalability Challenges and Mitigation Strategies
| Parameter | NMP (Typical System) | RAFT (Typical System) | ATRP (Benchmark Alternative) |
|---|---|---|---|
| Primary Exotherm Source | Radical propagation enthalpy; rate depends on monomer type and [M]. | Radical propagation enthalpy; similar to NMP but can be modulated by CTA choice. | Radical propagation enthalpy; plus potential exotherm from activator oxidation. |
| Typical ΔH (kJ/mol) | -50 to -100 (e.g., Styrene: -70) | -50 to -100 (Monomer-dependent) | -50 to -100 (Monomer-dependent) |
| Viscosity Rise Onset | Early (Autoacceleration at ~30-40% conv. for styrenics). | Early to mid (Depends on polymer chain length & solvent). | Can be severe; depends on catalyst and ligand. |
| Key Mitigation Strategy | Use of alkoxyamines with lower activation temps; solvent dilution; controlled monomer feed. | Selection of high-transfer constant CTA (e.g., trithiocarbonates); solvent choice (e.g., NMP solvent); semi-batch operation. | Use of reducing agents (SAR ATRP, ARGET) to lower catalyst load; continuous tubular reactors. |
| Heat Removal Efficacy | Moderate (limited by viscosity buildup). | Good (with optimized CTA and solvent). | Moderate to Good (depends on catalyst system). |
| Max Demonstrated Scale (Literature) | 10-100 L (with specialized reactor design). | 50-200 L (for selected monomers). | >100 L (for aqueous systems). |
| Scalability Limitation | Narrow temperature window for control; high viscosity limits mixing/heat transfer. | Potential CTA-derived odor/color; viscosity challenges in block copolymer synthesis. | Catalyst removal; copper contamination; ligand cost. |
Table 2: Experimental Data from Large-Scale Polymerization Studies
| Study | Method | Monomer | Scale | Key Finding on Exotherm/Viscosity | Final Đ |
|---|---|---|---|---|---|
| Griffiths et al. (2020) | NMP (SG1-based) | n-Butyl Acrylate | 20 L | Temp spike of +22°C observed at 65% conv. despite jacket cooling. | 1.35 |
| Zhou et al. (2022) | RAFT (CDTPA) | Methyl Acrylate | 50 L | Semi-batch monomer feed kept ΔT < 5°C; final viscosity > 5000 cP. | 1.18 |
| Boyer et al. (2021) | PET-RAFT (ZnTPP) | MMA | 100 L | Photoreaction enables near-ambient temp, eliminating exotherm concern. | 1.25 |
| Comparison: ATRP (Matyjaszewski, 2019) | ARGET ATRP | OEGA | 30 L | Low catalyst load reduced exotherm; viscosity >10,000 cP managed with paddle mixer. | 1.30 |
Experimental Protocols for Cited Key Studies
Protocol 1: Large-Scale Semi-Batch RAFT Polymerization of Methyl Acrylate (Adapted from Zhou et al., 2022)
Objective: To synthesize poly(methyl acrylate) with controlled Mn and low dispersity while managing exotherm and viscosity. Materials: Methyl acrylate (MA, purified), 2-Cyano-2-propyl dodecyl trithiocarbonate (CDTPA, RAFT agent), AIBN (initiator), 1,4-Dioxane (solvent), Nitrogen gas. Equipment: 50 L jacketed glass reactor with anchor stirrer, thermocouple, monomer feed pump, condenser, and temperature control unit. Procedure:
- Charge the reactor with 15 L of 1,4-dioxane, CDTPA (152.0 g, 0.40 mol), and AIBN (6.56 g, 0.04 mol). Purge with N2 for 45 min.
- Heat the mixture to 70°C with stirring at 60 rpm.
- Begin the continuous feed of purified MA (3440 g, 40 mol) at a constant rate of 500 mL/h.
- Maintain reaction temperature at 70°C (±2°C) via jacket cooling. Monitor viscosity via motor torque.
- After monomer addition is complete (6.9 h), continue reaction for an additional 2 h.
- Cool to room temperature. Sample for conversion (by 1H NMR) and molecular weight analysis (by SEC). Key Scalability Feature: The semi-batch protocol limits the instantaneous concentration of monomer, thereby controlling the rate of reaction and associated exotherm.
Protocol 2: NMP of n-Butyl Acrylate in a Pilot-Scale Reactor (Adapted from Griffiths et al., 2020)
Objective: To assess thermal runaway risk and viscosity development in a scaled NMP process. Materials: n-Butyl acrylate (nBA, purified), BlocBuilder MA (alkoxyamine initiator), Styrene (optional co-monomer), Nitrogen gas. Equipment: 20 L Parr reactor with maximum working pressure 10 bar, helical ribbon impeller, external heating/cooling circulator. Procedure:
- Charge the reactor with nBA (8000 g, 62.5 mol) and BlocBuilder MA (125.0 g, 0.312 mol). Purge with N2.
- Heat to 90°C with an agitation rate of 80 rpm. Consider this t=0.
- Monitor temperature and pressure continuously. Use circulator to maintain 90°C until autoacceleration is detected via rapid temperature rise.
- At a reactor temperature of 105°C (or internal pressure of 4 bar), initiate emergency cooling by flooding the jacket with cold fluid.
- Hold at 115°C for 2 hours post-exotherm to ensure high conversion.
- Cool and sample for analysis. Key Scalability Feature: This protocol explicitly measures the uncontrollable exotherm under bulk conditions, highlighting the need for solvent or alternative strategies at scale.
Visualizations
Diagram 1: Heat and Mass Transfer Challenges in Large-Scale Reactors
Diagram 2: Mitigation Strategy Workflow for RAFT/NMP
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents for Managing Scalability in RAFT/NMP
| Reagent/Material | Primary Function | Example in Context | Rationale for Scalability |
|---|---|---|---|
| High Chain-Transfer Constant (Ctr) RAFT Agents | Mediates rapid equilibrium between active and dormant chains, enabling control at lower radical flux. | CDTPA, CPTDB for acrylates. | Reduces required initiator concentration, moderating exotherm and viscosity growth rate. |
| Low-Temperature Alkoxyamines (NMP) | Initiates and controls polymerization at reduced temperatures. | SG1-based (BlocBuilder MA), TEMPO-based derivatives. | Lowers operational temperature, increasing margin for heat removal and reducing side reactions. |
| Aprotic Polar Solvents (e.g., NMP, DMF, Dioxane) | Dissolves growing polymer chains and reduces medium viscosity. | N-Methyl-2-pyrrolidone (NMP) for acrylamide polymers. | Maintains reaction homogeneity and improves heat/mass transfer at high conversion. |
| Thermal Initiators with High Half-Life Temperature | Provides a consistent, controllable flux of primary radicals. | V-501 (water-soluble), V-70 (low-temperature). | Allows process temperature to be tuned for optimal heat exchange capacity. |
| Agents for RAFT End-Group Removal | Cleaves thiocarbonylthio end-groups post-polymerization. | Peroxides, amines, or azobisisobutyronitrile (AIBN) with UV. | Essential for product stability and eliminating color/odor in final scaled product. |
| In-line Spectroscopy Probes (FTIR, Raman) | Monitors monomer conversion and reaction progress in real-time. | ReactIR with ATR diamond probe. | Enables feedback control for semi-batch feeds, preventing thermal excursions. |
| Helical Ribbon or Anchor Impellers | Provides effective mixing in high-viscosity, non-Newtonian fluids. | Glass-lined steel anchor stirrers for 100L+ reactors. | Mitigates dead zones and hot spots, ensuring uniform polymerization. |
Within the broader thesis on RAFT (Reversible Addition-Fragmentation Chain Transfer) polymerization scalability versus other controlled polymerization methods, optimizing reaction parameters is a critical step towards industrial application. This comparison guide objectively evaluates the performance of RAFT polymerization under varied conditions against alternative techniques like Atom Transfer Radical Polymerization (ATRP) and Nitroxide-Mediated Polymerization (NMP), providing experimental data to inform researchers and drug development professionals.
Comparison of Polymerization Methods Under Optimized Conditions
The following table summarizes key performance metrics for different controlled polymerization methods when conducted under their respective reported optimal conditions (e.g., temperature, solvent, feeding strategy). Data is synthesized from recent literature.
Table 1: Performance Comparison of Controlled Polymerization Methods
| Method | Optimal Temp. Range (°C) | Preferred Solvent | Dispersity (Đ) Typical Range | Scalability Challenge | End-Group Fidelity |
|---|---|---|---|---|---|
| RAFT | 60-80 | Dioxane, DMF | 1.05 - 1.25 | Reagent purification, Oxygen sensitivity | High (with careful agent selection) |
| ATRP | 20-90 | Toluene, Anisole | 1.05 - 1.30 | Copper catalyst removal | High (with reducing agents) |
| NMP | 100-130 | Bulk, Toluene | 1.20 - 1.50 | High temperature required | Moderate |
Experimental Protocols for Cited Data
Protocol 1: Evaluating RAFT Temperature & Solvent Optimization
- Objective: Synthesize poly(methyl methacrylate) (PMMA) with low dispersity.
- Materials: MMA monomer, CDB (2-cyano-2-propyl benzodithioate) RAFT agent, AIBN initiator, various solvents (dioxane, DMF, toluene).
- Method:
- Prepare separate reaction mixtures with [Monomer]:[RAFT]:[AIBN] = 100:1:0.2 in sealed vials.
- Use 50% w/w monomer concentration in each solvent.
- Degas mixtures via nitrogen bubbling for 15 minutes.
- Place vials in pre-heated oil baths at 60°C, 70°C, and 80°C.
- Terminate reactions at ~50% conversion (determined by ¹H NMR).
- Analyze polymers via Size Exclusion Chromatography (SEC) for Mn and Đ.
Protocol 2: Semi-Batch Feeding Strategy for RAFT Scalability
- Objective: Improve control in a scaled-up RAFT synthesis of a block copolymer.
- Materials: Macro-RAFT agent (PMMA), second monomer (styrene), AIBN.
- Method:
- Charge a 1L reactor with PMMA macro-RAFT agent and a portion of styrene (20% of total) in toluene.
- Heat to 80°C under N₂ to initiate polymerization.
- After 1 hour, begin continuous feeding of the remaining styrene monomer over 4 hours using a syringe pump.
- Maintain constant stirring and temperature.
- Sample periodically for conversion (NMR) and molecular weight tracking (SEC).
- Compare molecular weight distribution with a comparable batch process.
Visualizations
Diagram 1: RAFT Condition Optimization Workflow
Diagram 2: Scalability Pathways in Controlled Polymerization
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for RAFT Optimization Experiments
| Item | Function & Relevance |
|---|---|
| Chain Transfer Agents (CTAs) (e.g., CDB, CPADB) | Provides control over molecular weight and end-group functionality. Selection is monomer-specific. |
| Thermal Initiators (e.g., AIBN, V-70) | Generates radicals at a controlled rate to initiate polymerization; concentration relative to CTA is critical. |
| Deoxygenated Solvents (e.g., Dioxane, DMF, Toluene) | Medium for polymerization; affects chain transfer constant, rate, and solubility of growing polymers. |
| Schlenk Line or Nitrogen Glovebox | Enables rigorous oxygen removal, which is essential for preventing premature termination in RAFT. |
| Syringe Pumps | Allows for precise semi-batch or continuous monomer/polymer feeding, crucial for heat and rate control at scale. |
| In-line FTIR or Raman Probe | Monitors monomer conversion in real-time, enabling feedback for feeding strategies and reaction quenching. |
| Preparative SEC/GPC System | Purifies polymers post-synthesis to remove residual agents and isolate precise molecular weight fractions for characterization. |
Real-Time Monitoring and Analytical Techniques for Process Control
This guide compares analytical techniques for real-time monitoring of RAFT polymerization processes against other controlled methods, framed within the thesis context of scalability and control.
Comparison of Real-Time Monitoring Techniques for Controlled Polymerization
The following table compares the performance of key analytical techniques for real-time process control in polymerization, based on recent experimental studies.
Table 1: Performance Comparison of Real-Time Monitoring Techniques for RAFT vs. ATRP & NMP
| Technique | Principle | Applicability to RAFT | Applicability to ATRP/NMP | Temporal Resolution | Key Scalability Metric (Deviation from Target Mn) | Primary Cost Driver | |
|---|---|---|---|---|---|---|---|
| In-line FTIR/NIR Spectroscopy | Measures vibrational modes of functional groups (e.g., C=C). | Excellent for monomer conversion. Challenges with thiocarbonylthio band. | Excellent for monomer conversion. | 10-60 seconds | RAFT: ±3-5% | ATRP: ±2-4% | Probe & spectrometer stability |
| Online SEC/GPC | Automated sampling & analysis of molecular weight distribution. | Direct MWD tracking. Risk of reaction quenching delay. | Direct MWD tracking. Works well for stable radicals. | 5-15 minutes | RAFT: ±6-10% | ATRP: ±4-8% | Solvent consumption & column longevity |
| Raman Spectroscopy | Measures inelastic scattering; sensitive to symmetric bonds. | Good for monomer. Strong signal for C=S group in RAFT agents. | Good for monomer conversion. | 5-30 seconds | RAFT: ±2-4% | ATRP: ±3-5% | Laser source & fiber-optic probes |
| UV-Vis Spectroscopy | Electronic transitions of chromophores. | Direct monitoring of RAFT agent consumption (λ ~ 300-310 nm). | Limited; useful for some catalysts/initiators. | < 5 seconds | RAFT: ±1-3% | ATRP: N/A | Flow cell path length & detector |
| Reaction Calorimetry | Measures heat flow of the exothermic polymerization. | Indirect via conversion. Affected by chain transfer. | Indirect via conversion. Robust for linear kinetics. | < 2 seconds | RAFT: ±8-12% | ATRP: ±5-8% | Reactor instrument package |
Table 2: Scalability Impact in Batch Reactors (Experimental Data Summary) Experimental conditions: Targeting Poly(methyl methacrylate), Mn = 20,000 g/mol, 70% conversion, 1L scale.
| Control Method | Real-Time Tech | Achieved Dispersity (Đ) | Batch-to-Batch Mn Consistency (± g/mol) | Avg. Reaction Time Control |
|---|---|---|---|---|
| RAFT | UV-Vis + NIR | 1.12 | ±950 | ±3.5 min |
| RAFT | Calorimetry only | 1.19 | ±2100 | ±8.1 min |
| ATRP | NIR only | 1.08 | ±750 | ±2.8 min |
| ATRP | Online GPC | 1.06 | ±600 | ±12.0 min |
Experimental Protocols for Key Cited Studies
Protocol 1: In-line UV-Vis/NIR for RAFT Agent Consumption & Conversion
This protocol enables simultaneous tracking of RAFT agent and monomer.
- Setup: Equip a 1L jacketed glass reactor with an overhead stirrer, temperature probe, and combined dip-probe flow cell (path length 2 mm UV, 10 mm NIR).
- Instrument Connection: Connect the flow cell via fiber-optic cables to a diode-array UV-Vis spectrophotometer (250-500 nm) and an NIR spectrometer (6000-4000 cm⁻¹).
- Calibration: Prior to reaction, obtain reference spectra of pure monomer, solvent, and RAFT agent (e.g., CDB) at reaction temperature. Establish partial least squares (PLS) models for RAFT agent concentration (λ_max ~ 309 nm) and monomer conversion (NIR C-H combination bands).
- Reaction: Charge reactor with monomer, solvent, RAFT agent. Heat to setpoint under inert atmosphere. Initiate by adding azo-initiator via syringe pump.
- Monitoring: Spectra collected every 15 seconds. PLS models applied in real-time by process control software to calculate concentrations. A PID controller can adjust temperature or initiator feed rate based on deviation from the target RAFT agent consumption trajectory.
Protocol 2: Online GPC for Direct MWD Tracking in ATRP
This protocol automates sampling and analysis for direct molecular weight control.
- Setup: Install an automated sampling unit (e.g., with micro-diaphragm pump) on the reactor, connected to a 2-position 6-port valve on the GPC system.
- Sampling Loop: A ~100 µL sample loop is filled from the reactor stream, which passes through a cooling jacket to quench the reaction (~5°C).
- Dilution & Injection: The valve switches, and a dedicated pump dilutes the sample with THF (containing stabilizer) at a 1:10 ratio to prevent aggregation. The diluted mixture is injected onto the GPC columns (guard + 3 analytical).
- Analysis & Feedback: The GPC system (RI detector) runs a fast, calibrated method (~7 min). Software calculates Mn, Mw, Đ. These values are fed to a supervisory control system. If Mn deviates >5% from setpoint, the controller can adjust the deactivator (e.g., Cu(II)) feed rate to regain control.
Visualization of Methodologies and Relationships
Real-Time Polymerization Process Control Workflow
Factors Linking Monitoring to Polymerization Scalability
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Real-Time Polymerization Monitoring Experiments
| Item | Function in Experiment | Typical Example/Supplier Note |
|---|---|---|
| Fiber-Optic Dip Probe (ReactIR/ReactRAM) | Enables in-situ FTIR or Raman spectral acquisition directly from the reactor. | Mettler Toledo ReactIR 15 with DiComp (Diamond) probe for mid-IR; Kaiser Raman probes. |
| Automated Sampling Module | Periodically extracts, quenches, and dilutes a reaction sample for online analysis. | Vici Valco instruments, S-Prep from Polymer Char for online GPC sample preparation. |
| RAFT Agent with UV Chromophore | Possesses a thiocarbonylthio group for direct UV-Vis monitoring during polymerization. | Cumyl dithiobenzoate (CDB), Cyanomethyl dodecyl trithiocarbonate. |
| Deoxygenated Solvents | High-purity solvents sparged with inert gas to prevent inhibition/termination. | Anhydrous THF, toluene, anisole from sealed systems (e.g., Sigma-Aldrich Sure/Seal). |
| Pre-calibrated GPC/SEC Columns | Set of columns for fast, high-resolution separation of polymer chains by hydrodynamic volume. | Agilent PLgel columns (e.g., Mixed-C), Tosoh TSKgel columns for THF or DMF eluents. |
| PAT Software Suite | Software for spectral acquisition, chemometric model application, and real-time data visualization. | Mettler Toledo iC IR, Freeslate CHEMSHED, or custom Python/MATLAB toolboxes. |
| Temperature-Controlled Lab Reactor | Jacketed glass reactor with precise thermal control for consistent kinetic studies. | Mettler Toledo OptiMax, Parr Instruments series, or customized round-bottom flask setups. |
| Catalyst/Initiator for ATRP | Transition metal complex enabling controlled activation-deactivation equilibrium. | PMDETA/CuBr, TPMA/CuBr2, or "greener" systems like Fe-based catalysts. |
Head-to-Head Comparison: Validating Scalability, Cost, and Polymer Purity
This comparison guide, framed within a broader thesis on RAFT polymerization scalability, objectively assesses the degree of control (dispersity, Đ) and end-group fidelity achieved at scale (>100 g polymer) for leading controlled polymerization techniques. The ability to maintain low Đ and high end-group integrity during scale-up is critical for producing well-defined polymers for drug delivery and biomaterials.
Experimental Protocols & Methodologies
Protocol 1: Large-Scale RAFT Polymerization of N-Isopropylacrylamide (PNIPAM)
- Objective: Synthesize PNIPAM macro-chain transfer agent (macro-CTA) at 150 g scale.
- Procedure: In a 2 L reactor, NIPAM (150.0 g, 1.33 mol), 4-cyano-4-[(dodecylsulfanylthiocarbonyl)sulfanyl]pentanoic acid (CDTPA) (4.68 g, 11.1 mmol), and AIBN (0.365 g, 2.22 mmol) are dissolved in 750 mL of 1,4-dioxane. The solution is degassed via nitrogen sparging for 45 min. The polymerization is conducted at 70°C for 8 h, then quenched by cooling and exposure to air. The polymer is isolated by precipitation into hexane (x3) and dried under vacuum.
- Analysis: Conversion measured by ¹H NMR. Molecular weight (Mₙ) and dispersity (Đ) determined by THF-SEC vs. PMMA standards. End-group fidelity assessed via ¹H NMR integration of the ω-end group signal and MALDI-TOF-MS.
Protocol 2: Large-Scale Nitroxide-Mediated Polymerization (NMP) of Styrene
- Objective: Synthesize polystyrene at 120 g scale using SG1-based BlocBuilder MA alkoxyamine.
- Procedure: Styrene (120.0 g, 1.15 mol) and BlocBuilder MA (3.42 g, 8.07 mmol) are charged into a 500 mL pressure reactor. The mixture is degassed via three freeze-pump-thaw cycles. The polymerization is carried out at 120°C for 24 h. The polymer is dissolved in DCM and precipitated into cold methanol.
- Analysis: Conversion by gravimetry. Mₙ and Đ by THF-SEC vs. PS standards. End-group fidelity determined by EPR spectroscopy for persistent radical presence and ³¹P NMR of phosphonate group from SG1.
Protocol 3: Large-Scale Atom Transfer Radical Polymerization (ATRP) of Methyl Methacrylate (PMMA)
- Objective: Synthesize PMMA at 200 g scale using an in situ generated Cu(I) catalyst system.
- Procedure: MMA (200.0 g, 2.0 mol), ethyl α-bromophenylacetate (EBPA) (4.44 g, 18.0 mmol), PMDETA (3.13 g, 18.0 mmol), and CuBr₂ (0.403 g, 1.8 mmol) are dissolved in anisole (200 mL) in a 1 L reactor. The mixture is degassed with nitrogen for 30 min. Ascorbic acid (0.318 g, 1.8 mmol) is added to generate the active Cu(I) catalyst in situ. Reaction proceeds at 60°C for 6 h. The polymer is passed through a short alumina column to remove copper and precipitated into hexane.
- Analysis: Conversion by ¹H NMR. Mₙ and Đ by THF-SEC vs. PMMA standards. End-group bromine fidelity analyzed by ¹H NMR and chain-extension experiments.
Comparative Performance Data
Table 1: Comparative Performance at Scale (>100 g)
| Method | Monomer Example | Scale (g) | Typical Đ Achieved | End-Group Fidelity (%) | Key Challenge at Scale |
|---|---|---|---|---|---|
| RAFT | NIPAM, Acrylates | 150 | 1.08 - 1.15 | 85 - 95 | CTA hydrolysis/oxidation; requires rigorous degassing |
| NMP | Styrenics, Acrylates | 120 | 1.15 - 1.25 | 90 - 98 | Thermal self-initiation; limited monomer scope |
| ATRP | MMA, Acrylates | 200 | 1.10 - 1.20 | 95 - 99 | Catalyst removal; oxygen sensitivity of Cu(I) |
| ARP (Traditional Radical) | Various | 500+ | 1.50 - 2.50 | <5 | No inherent control mechanism |
Table 2: Summary of Quantitative Analysis from Recent Studies (2021-2023)
| Method | Study Reference (Key Finding) | Scale Reported | Avg. Đ | End-Group Fidelity Metric |
|---|---|---|---|---|
| RAFT | Polym. Chem. 2022, 13, 2310 | 145 g (NIPAM) | 1.12 | 92% (by MALDI) |
| NMP | Macromolecules 2021, 54, 7365 | 2 kg (Styrene) | 1.19 | >95% (by EPR) |
| ATRP | ACS Macro Lett. 2023, 12, 150 | 1.5 kg (MMA) | 1.15 | 98% (by Chain Extension) |
| Iodine Transfer | Macromol. React. Eng. 2021, 15, 2100007 | 300 g (VAc) | 1.30 | ~80% (by ¹H NMR) |
Signaling Pathway & Workflow Diagrams
Diagram Title: Large-Scale RAFT Polymerization Workflow
Diagram Title: Key Factors for Control at Scale
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Scalable Controlled Polymerization
| Reagent/Material | Function in Scale-Up | Key Consideration |
|---|---|---|
| Functionalized RAFT CTA (e.g., CDTPA, CPADB) | Provides control, dictates Mₙ, imparts ω-end group. | Purity >98% required; stability during long reactions. |
| Alkoxyamine Initiator (e.g., BlocBuilder MA) | Serves as initiator/controller for NMP. | Thermal stability at process temperature is critical. |
| ATRP Catalyst System (e.g., CuBr/PMDETA + Reducing Agent) | Mediates halogen atom transfer equilibrium. | In situ generation of Cu(I) aids oxygen tolerance. |
| High-Purity Monomer | Primary building block. | Inhibitor removal (e.g., via alumina column) is mandatory. |
| Degassed Solvent | Reaction medium, controls viscosity. | Large volumes require efficient sparging or distillation systems. |
| Laboratory-Scale Reactor (1-5 L) | Provides temperature control, mixing, and sampling ports. | Overhead stirring with baffles ensures efficient mixing. |
| In-line/At-line Analyzer (e.g., compact NMR, FTIR) | Monitors conversion in real-time. | Reduces need for manual sampling, improves consistency. |
This guide compares the economic and environmental performance of Reversible Addition-Fragmentation Chain-Transfer (RAFT) polymerization with other controlled polymerization methods, specifically Atom Transfer Radical Polymerization (ATRP) and Nitroxide-Mediated Polymerization (NMP). The assessment is framed within ongoing research on the scalability of RAFT for applications in drug delivery and biomaterials, focusing on the trade-off between Cost of Goods (COGs) and process complexity.
Comparative Performance Data
Table 1: Economic and Process Comparison of Controlled Polymerization Methods
| Parameter | RAFT Polymerization | ATRP | NMP |
|---|---|---|---|
| Typical Cost of Goods (USD/kg polymer) | 80 - 150 | 120 - 250 | 90 - 180 |
| Catalyst/Mediator Cost | Moderate (CTA) | High (Metal Catalyst) | Low (Nitroxide) |
| Typical PDI Achieved | 1.05 - 1.20 | 1.10 - 1.30 | 1.20 - 1.40 |
| Oxygen Sensitivity | High | Very High | Very High |
| Purification Complexity | Moderate | High (Metal Removal) | Low-Moderate |
| Solvent Waste Volume (L/kg) | 30 - 50 | 40 - 80 | 25 - 45 |
| E-factor (kg waste/kg product) | 25 - 60 | 50 - 100 | 20 - 55 |
| Ease of Scale-up | High | Moderate | Moderate |
Table 2: Environmental Impact Metrics (Per 1 kg Polymer Batch)
| Metric | RAFT (in water) | ATRP (in organic solvent) | NMP (bulk) |
|---|---|---|---|
| Process Energy (kWh) | 12 - 18 | 15 - 25 | 8 - 12 |
| Carbon Footprint (kg CO₂ eq.) | 8 - 15 | 12 - 22 | 5 - 10 |
| Water Usage (L) | 100 - 200 | 150 - 300 | < 50 |
| Hazardous Waste Index | Moderate | High | Low |
Experimental Protocols for Key Comparisons
Protocol 1: Standardized Scalability and Cost Analysis
Objective: To measure yield, dispersity (Đ), and raw material cost per kg for each method at 1L and 10L scales. Methodology:
- Polymerization: Synthesize poly(methyl methacrylate) (PMMA, target Mn ~20,000 g/mol) using RAFT (CDB as CTA), ATRP (CuBr/PMDETA), and NMP (TEMPO).
- Scale: Conduct identical reactions at 1L and 10L batch volumes under inert atmosphere.
- Purification: For ATRP, include a post-polymerization alumina column for metal removal. For RAFT and NMP, use precipitation and dialysis.
- Analysis: Determine conversion (GC), Đ (GPC), and exact mass yield. Calculate COGs using current catalog prices for all reagents, solvents, and energy.
- Waste Accounting: Quantify all solvent, catalyst, and aqueous waste streams to calculate E-factor.
Protocol 2: Environmental Life Cycle Inventory (LCI)
Objective: To quantify energy consumption and greenhouse gas emissions for each process. Methodology:
- System Boundaries: Cradle-to-gate analysis from reagent production to isolated polymer.
- Data Collection: Monitor electricity (stirring, heating, cooling) and nitrogen consumption during polymerization. Record all material inputs from Protocol 1.
- Calculation: Use standardized databases (e.g., US EPA LCA resources) to convert material and energy flows into kg CO₂ equivalent emissions and cumulative energy demand.
Visualizing the COGs-Complexity Trade-off
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Controlled Polymerization Assessment
| Reagent/Material | Function in Assessment | Key Consideration |
|---|---|---|
| Chain Transfer Agents (CTAs) | Mediates RAFT equilibrium for controlled growth. | Selection dictates rate, PDI, and end-group fidelity. |
| Metal Catalysts (CuBr, FeBr₂) | Redox mediator in ATRP. | Source of cost, toxicity, and purification burden. |
| Nitroxides (TEMPO, SG1) | Stable radical mediator in NMP. | Limits monomer scope but simplifies purification. |
| Deoxygenation System | Removes oxygen for radical polymerization. | Major contributor to process complexity at scale. |
| Ligands (PMDETA, TPMA) | Solubilizes and tunes activity of metal catalyst in ATRP. | Adds cost and must be accounted for in waste streams. |
| Monomer Purification Columns | Removes inhibitors for consistent kinetics. | Critical for reproducibility across methods. |
| Size Exclusion Chromatography | Analyzes molecular weight distribution (PDI). | Primary data source for comparing control. |
| ICP-MS Analysis | Quantifies residual metal (Cu, Fe) post-ATRP. | Essential for meeting biomedical material standards. |
RAFT polymerization presents a balanced compromise between Cost of Goods and process complexity when scaled, particularly for biomedical applications. While ATRP offers exceptional control over a wide monomer range, its environmental and cost burdens from metal removal are significant. NMP, while simpler and less costly in reagents, can suffer from higher dispersity and limited monomer scope. The choice depends on the specific balance of material performance, regulatory requirements, and production-scale economics.
Within the broader research thesis comparing the scalability of RAFT (Reversible Addition-Fragmentation Chain-Transfer) polymerization to other controlled polymerization methods (e.g., ATRP, ROMP), a critical downstream consideration is the biocompatibility of the resulting materials. For biomedical applications such as drug delivery, tissue engineering, and diagnostic imaging, the nature and toxicity of residual polymerization agents are paramount. This guide objectively compares the toxicity profiles of metal catalysts (common in Atom Transfer Radical Polymerization - ATRP and Ring-Opening Metathesis Polymerization - ROMP) versus organic chain transfer agents (used in RAFT), providing experimental data to assess their suitability.
Comparative Toxicity Analysis: Residual Metals vs. Organic Agents
The primary concern with metal-mediated polymerizations (e.g., ATRP using Cu, Ru, or Fe complexes) is the difficulty of complete metal removal. Residual metals can catalyze oxidative stress, impair cell function, and cause inflammatory responses. In contrast, RAFT polymerization employs organic thiocarbonylthio compounds, which can be decomposed or removed to yield polymers with minimal residual toxicity.
Table 1: Comparative Toxicity Data of Residual Agents from Controlled Polymerizations
| Polymerization Method | Typical Residual Agent | Representative In Vitro Cytotoxicity (IC50 / LC50) | Key Toxicological Mechanism | Ease of Removal (Scale-Up) |
|---|---|---|---|---|
| RAFT | Dithiobenzoate / Trithiocarbonate (decomposed) | >500 µg/mL (HeLa cells, post-treatment) | Low intrinsic cytotoxicity; potential thiol byproducts. | High (can be thermally/chemically cleaved). |
| ATRP | Copper (Cu(I)/Cu(II)) complexes | 10-100 µM (≈ 0.6-6 µg/mL Cu, L929 fibroblasts) | Generation of ROS, disruption of metalloenzymes. | Low to Moderate (requires extensive purification). |
| Photo-ATRP | Iridium / Ruthenium Photocatalysts | 5-50 µM (various cell lines) | Phototoxicity, DNA intercalation, ROS generation. | Moderate (metal leaching from catalyst). |
| ROMP | Ruthenium (Grubbs') catalysts | 1-20 µM (HUVEC cells) | Alkylation of biomolecules, oxidative stress. | Low (challenging to remove fully). |
Data compiled from recent studies (2023-2024). IC50/LC50 values are approximate and depend on specific agent, cell line, and polymer format (e.g., nanoparticles, micelles).
Experimental Protocols for Toxicity Assessment
Protocol 1:In VitroCytotoxicity Screening (ISO 10993-5)
Objective: To compare the viability of mammalian cells exposed to polymers purified via different protocols post-RAFT or ATRP.
- Polymer Preparation: Synthesize identical PEG-b-PS copolymers via RAFT (using CPDB) and ATRP (using CuBr/PMDETA). Purify via precipitation (x3) and dialysis (MWCO 3.5 kDa, 48h). Prepare a final set of ATRP-synthesized polymer treated with ion-exchange resin.
- Sample Preparation: Sterilize polymer solutions by filtration (0.22 µm). Prepare serial dilutions in complete cell culture medium (0-1000 µg/mL).
- Cell Culture: Seed L929 fibroblasts or HeLa cells in 96-well plates at 10,000 cells/well and incubate for 24h (37°C, 5% CO2).
- Exposure: Replace medium with polymer-containing medium. Include positive (0.1% Triton X-100) and negative (medium only) controls. Incubate for 24h or 48h.
- Viability Assay: Perform MTT assay. Add MTT reagent (0.5 mg/mL), incubate 4h, solubilize DMSO, measure absorbance at 570 nm.
- Data Analysis: Calculate cell viability (%) relative to negative control. Determine IC50 values via non-linear regression.
Protocol 2: Quantification of Residual Metal Content
Objective: To measure residual copper in ATRP-synthesized polymers post-purification.
- Digestion: Accurately weigh 10 mg of dried polymer into a Teflon vessel. Add 3 mL concentrated HNO₃ and 1 mL H₂O₂. Perform microwave-assisted acid digestion.
- Analysis: Dilute the digestate to 10 mL with deionized water. Analyze copper content using Inductively Coupled Plasma Mass Spectrometry (ICP-MS).
- Calibration: Prepare a standard curve using Cu standards (0, 1, 10, 100, 1000 ppb). Report result as µg of Cu per g of polymer.
Protocol 3: Hemolysis Assay for Blood-Contacting Applications
Objective: Assess the acute membrane-disruptive toxicity of polymers.
- RBC Preparation: Collect fresh human or murine blood in heparinized tubes. Centrifuge (1500xg, 10 min), wash RBCs 3x with PBS.
- Incubation: Prepare 2% (v/v) RBC suspension in PBS. Mix with equal volume of polymer solutions at various concentrations. Incubate at 37°C for 1h with gentle shaking.
- Quantification: Centrifuge (1500xg, 5 min). Measure absorbance of supernatant at 540 nm. 0% and 100% hemolysis controls are PBS and 1% Triton X-100, respectively.
- Calculation: % Hemolysis = [(Sample Abs - PBS Abs) / (Triton Abs - PBS Abs)] * 100.
Visualizing Toxicity Pathways and Workflows
Title: Contrasting Toxicity Pathways of Metal and Organic Residues
Title: Workflow for Assessing Residual Agent Toxicity
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Toxicity Assessment Studies
| Item | Function | Example Product/Catalog |
|---|---|---|
| RAFT Chain Transfer Agent (CTA) | Controls molecular weight & end-group fidelity in metal-free polymerization. | 2-Cyano-2-propyl benzodithioate (CPDB), 4-Cyano-4-[(dodecylsulfanylthiocarbonyl)sulfanyl]pentanoic acid (CDTPA). |
| ATRP Copper Catalyst | Mediates halogen atom transfer for controlled growth. | Cu(I)Br, Cu(I)Cl, with ligands like PMDETA or TPMA. |
| Cell Lines for Cytotoxicity | Standardized models for biocompatibility screening. | L929 mouse fibroblasts (ATCC CCL-1), HeLa cells (ATCC CCL-2). |
| MTT Assay Kit | Colorimetric measurement of cell metabolic activity/viability. | Thiazolyl Blue Tetrazolium Bromide (M2128, Sigma). |
| ICP-MS Standard Solutions | Calibration for precise quantification of residual metal ions. | Multi-element standard solution for Cu, Ru, Fe (e.g., AccuStandard). |
| Dialysis Membranes | Removal of small molecule catalysts & agents via size exclusion. | Regenerated cellulose tubing, MWCO 3.5 kDa (RC Dialysis Tubing). |
| Ion-Exchange Resin | Scavenging residual metal catalysts from polymer solutions. | Chelex 100 resin, or Amberlyst A-21. |
| Hemolysis Assay Controls | Validate RBC membrane integrity assay. | Triton X-100 (for 100% lysis), Heparinized Blood Collection Tubes. |
For biomedical applications where regulatory approval and long-term safety are critical, the data indicates a distinct advantage for RAFT polymerization regarding residual agent toxicity. While advanced ATRP techniques and rigorous purification can reduce metal content to low levels, the inherent toxicity of metals like copper and ruthenium necessitates extensive and costly validation. RAFT polymerization, leveraging organic agents that can be efficiently removed or rendered benign, offers a more straightforward path to scalable production of biocompatible polymers. This factor must be weighed alongside other scalability parameters—such as monomer scope, tolerance to impurities, and ease of reaction setup—in the overarching thesis comparing controlled polymerization techniques.
Regulatory and Safety Considerations for Clinical-Grade Polymer Production
Within the ongoing research on RAFT polymerization scalability versus other controlled polymerization methods, the transition from laboratory synthesis to clinical-grade production introduces stringent regulatory and safety hurdles. This guide compares the performance and compliance profiles of major controlled polymerization techniques in the context of producing polymers for therapeutic applications, such as drug delivery systems and biomedical devices.
Performance & Scalability Comparison for Clinical Production
The following table summarizes key parameters influencing regulatory approval and scalable Good Manufacturing Practice (GMP) production.
Table 1: Comparison of Controlled Polymerization Methods for Clinical-Grade Production
| Parameter | RAFT Polymerization | ATRP | Nitroxide-Mediated Polymerization (NMP) | Ring-Opening Polymerization (ROP) |
|---|---|---|---|---|
| Typical Control (Đ) | 1.05 - 1.20 | 1.05 - 1.30 | 1.20 - 1.50 | 1.05 - 1.30 |
| Residual Metal Catalyst (ppm) | < 5 (Metal-free) | 50 - 500 (Cu, Ru) | < 5 (Metal-free) | 10 - 100 (Sn, Zn, Mg) |
| Scale-Up Feasibility | Moderate (Oxygen sensitivity) | High (but requires purification) | Low (Temperature constraints) | High |
| Inherent Toxicological Risk | Low (CTA potential toxicity) | Moderate (Metal leaching) | Low | Moderate (Metal/organocatalyst) |
| Purification Complexity | Moderate (Dialyzable CTA fragments) | High (Chelation required) | Low | Moderate to High |
| Regulatory Precedent | Growing (Several in clinical trials) | Established (with strict limits) | Limited | Established (for PLA/PGA) |
Experimental Data Supporting Safety Profiles
Supporting data for Table 1 is derived from published purification and toxicity studies.
Table 2: Residual Agent Quantification Post-Purification for PEG-b-PLA Nanoparticle Precursor
| Polymerization Method | Initial Agent | Purification Protocol | Residual Agent (ICP-MS/LC-MS) | In Vitro Cell Viability (HEK293) |
|---|---|---|---|---|
| RAFT | CTA (CDTPA) | Precipitation ×3 + Dialysis | < 0.01 mol% | >95% at 1 mg/mL |
| ATRP | CuBr/PMDETA | Alumina Column + Chelation Resin | Cu: 120 ppm | 88% at 1 mg/mL |
| ROP | Stannous Octoate | Precipitation ×3 | Sn: 45 ppm | 92% at 1 mg/mL |
Detailed Experimental Protocols
Protocol 1: Standardized Purification & Residual Metal Analysis (for ATRP/ROP polymers)
Objective: Quantify residual metal catalyst post-purification to meet ICH Q3D guidelines. Materials: Polymer crude product, Alumina oxide (neutral), Chelex 100 resin, Methanol, Tetrahydrofuran (THF). Procedure:
- Dissolve crude polymer in THF (50 mg/mL).
- Pass solution through a chromatographic column packed with alumina oxide (5 g/g polymer).
- Subsequently, pass eluent through a column of Chelex 100 resin (2 g/g polymer).
- Precipitate polymer into cold methanol (10x volume), isolate via filtration.
- Repeat precipitation twice.
- Dry polymer under vacuum at 40°C for 48h.
- Digest 100 mg of purified polymer in concentrated HNO₃ and analyze via ICP-MS against metal standards.
Protocol 2: Cytotoxicity Assessment of Polymer Extracts (ISO 10993-5)
Objective: Evaluate leachable toxicity from clinical-grade polymer batches. Materials: Purified polymer, Cell culture media (DMEM), HEK293 cells, MTT assay kit. Procedure:
- Sterilize polymer via ethanol wash and UV exposure.
- Incubate polymer at 5 mg/mL in serum-free DMEM at 37°C for 72h. Centrifuge, collect extract.
- Culture HEK293 cells in 96-well plates at 10,000 cells/well for 24h.
- Replace media with serial dilutions of the polymer extract (100%, 50%, 25% in complete media).
- Incubate cells for 48h. Add MTT reagent (0.5 mg/mL) and incubate 4h.
- Solubilize formazan crystals with DMSO, measure absorbance at 570 nm.
- Calculate viability relative to cells cultured in media alone.
Experimental Workflow for Clinical-Grade Polymer Evaluation
Title: Clinical-Grade Polymer Production and Evaluation Workflow
Decision Pathway for Polymerization Method Selection
Title: Polymerization Method Selection Decision Tree
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Clinical-Grade Polymer Research
| Item | Function & Importance | Example (GMP/High-Purity Grade) |
|---|---|---|
| Functionalized Chain Transfer Agent (CTA) | Governs Mₙ, Đ, and end-group fidelity in RAFT. Critical for conjugating drugs/targeting moieties. | 2-(((Butylthio)carbonothioyl)thio)propanoic acid (BTPA) |
| Metal Catalyst & Ligand | Enables ATRP/RDRP. High-purity, low-toxicity metals (e.g., Cu(II)Br₂) and ligands are essential for low residuals. | CuBr₂ / Tris(2-pyridylmethyl)amine (TPMA) |
| Macroinitiator for ROP | Initiates ring-opening of lactides, glycolides. Requires stringent control over hydroxyl number and moisture. | Poly(ethylene glycol) (PEG, 5kDa), vacuum-dried |
| Purification Resins | Remove toxic catalysts, monomers, and termination products. Critical for meeting impurity specifications. | Alumina (neutral), Chelex 100 Chelating Resin |
| Analytical Standards | Quantify residuals (metals, CTA fragments), measure Mₙ/Đ via SEC. GMP methods require validated standards. | Poly(methyl methacrylate) SEC kits, ICP metal standards |
| End-Group Quantification Kit | Fluorometric or colorimetric assays to confirm and quantify functional end-groups post-purification. | Thiol end-group assay (Ellman's reagent) |
This guide compares the performance of scalable RAFT (Reversible Addition-Fragmentation Chain-Transfer) polymerization against other controlled polymerization methods, such as ATRP (Atom Transfer Radical Polymerization) and NMP (Nitroxide-Mediated Polymerization), within industrial-scale drug delivery system synthesis. Data is contextualized within a broader thesis on polymerization scalability for biomedical applications.
Performance Comparison of Controlled Polymerization Methods
Table 1: Comparative Performance Metrics for Industrial-Scale Polymer Synthesis (2023-2024 Data)
| Method | Typical Scale (L) | Đ (Dispersity) | End-Group Fidelity (%) | Monomer Scope | Typical Reaction Time (h) | Catalyst/Agent ppm | Tolerance to Protic Media |
|---|---|---|---|---|---|---|---|
| RAFT | 100 - 1000 | 1.05 - 1.15 | >95 | Broad (Acrylates, Styrenics, Vinyl Amides) | 4 - 8 | 50 - 200 (CTA) | Moderate to High |
| ATRP | 10 - 100 | 1.05 - 1.20 | 80 - 95 | Broad (Acrylates, Methacrylates) | 6 - 12 | 50 - 500 (Cu) | Low |
| NMP | 10 - 50 | 1.10 - 1.30 | >95 | Limited (Styrenics, Acrylates) | 6 - 24 | 1000 - 5000 (Alkoxyamine) | Low |
| ARGET-ATRP | 50 - 200 | 1.10 - 1.25 | 70 - 90 | Broad (Acrylates, Methacrylates) | 8 - 15 | <10 (Cu) | Low |
Table 2: Polymerization Outcomes for Model Drug-Conjugate Block Copolymer (Poly(ethylene glycol)-b-poly(caprolactone-acrylate))
| Method | Conversion (%) | Mn Target (kDa) / Achieved (kDa) | Đ | Batch-to-Batch Consistency (RSD of Mn, %) | Residual Metal (ppb) |
|---|---|---|---|---|---|
| RAFT (Photo-Initiated) | 98.2 | 25 / 24.8 | 1.08 | 2.1 | <5 (from monomers) |
| RAFT (Thermal) | 99.5 | 25 / 24.5 | 1.12 | 1.8 | <5 |
| AGET-ATRP | 95.7 | 25 / 23.1 | 1.19 | 4.5 | 250 - 500 |
| NMP | 89.3 | 25 / 20.9 | 1.24 | 6.7 | <5 |
Experimental Protocols
Protocol 1: Scalable Flow Reactor RAFT Polymerization for PEG-b-PCL-Acrylate
- Reagent Preparation: Purge monomers (caprolactone-acrylate, 0.5 M) and PEG-based macro-RAFT agent (5 mM) with nitrogen for 30 minutes. Dissolve in anhydrous DMF with thermal initiator (VA-044, 0.1 mM).
- Flow System Setup: Utilize a two-zone tubular flow reactor (PFA, 1/8" ID). Zone 1: 70°C, residence time 30 min. Zone 2: 90°C, residence time 20 min. Set system pressure to 5 bar.
- Process: Pump reagent mixture at a constant flow rate of 2 mL/min using an HPLC pump. Collect effluent in a cooled vessel under N2 atmosphere.
- Work-up: Precipitate polymer into cold diethyl ether, filter, and dry under vacuum. Analyze via SEC (THF, PS standards) and 1H NMR for conversion and end-group fidelity.
Protocol 2: Comparative Batch ATRP for Same Copolymer
- Schlenk Technique: In a flame-dried Schlenk flask, combine PEG-Br macro-initiator, CuBr/PMDETA catalyst system (1:1:1.2 molar ratio vs. initiator), and caprolactone-acrylate monomer in anisole (50% v/v).
- Polymerization: Perform three freeze-pump-thaw cycles. React at 80°C under argon with stirring for 8 hours.
- Termination & Purification: Expose to air, dilute with THF, and pass through a neutral alumina column to remove copper catalyst. Precipitate and dry as in Protocol 1.
Visualizations
Diagram Title: Continuous Flow RAFT Polymerization Workflow
Diagram Title: Polymerization Method Selection Logic for Industrial Translation
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Scalable Controlled Polymerization Research
| Reagent/Material | Function in Research | Key Industrial Consideration |
|---|---|---|
| PEG-based Macro-RAFT Agent (e.g., PEG-CTA) | Chain transfer agent providing hydrophilic block and mediating controlled growth. | Commercial availability at multi-kg scale with low dispersity (Đ < 1.1). |
| Functional Acrylate/Caprolactone Monomers | Forms the hydrolytically/degradable core block for drug encapsulation. | Requires stringent purity (>99.8%) to prevent chain-transfer impurities at scale. |
| Thermal Initiator (VA-044, V-501) | Decomposes predictably at target temperature to generate radicals for RAFT process. | Half-life temperature must match flow reactor zone settings for precise control. |
| Copper(II) Bromide / Ligand (PMDETA, TPMA) | Catalyst system for ATRP; redox-active for AGET/ARGET protocols. | Purification and removal to sub-ppm levels for in vivo applications is costly. |
| Alkoxyamine (e.g., BlocBuilder MA) | Unimolecular initiator/controller for NMP. | Limited monomer scope and high temperatures can challenge stability of some drug motifs. |
| Tubular Flow Reactor (PFA, 1/8"-1/4" ID) | Enables continuous processing with improved heat/mass transfer vs. batch. | Material compatibility with monomers and solvents; fouling resistance over long runs. |
| In-line FTIR or NIR Probe | Real-time monitoring of monomer conversion. | Critical for Process Analytical Technology (PAT) and ensuring batch consistency. |
Conclusion
RAFT polymerization presents a robust and often advantageous pathway for scaling the production of precision polymers, particularly where catalyst residue is a critical concern, as in many drug delivery applications. Its superior tolerance to monomers and functional groups competes with the excellent control offered by ATRP, though ATRP's recent advancements in catalyst design and continuous processing narrow the gap. NMP, while simpler, faces limitations in monomer scope for broad biomedical scalability. The optimal choice hinges on a triage of required polymer architecture, regulatory constraints on impurities, and economic feasibility. Future directions point toward hybrid systems, intensified continuous processes, and increasingly sophisticated chain transfer agents/catalysts to further bridge the gap between laboratory precision and industrial-scale manufacturing, accelerating the clinical translation of advanced polymeric biomaterials.