Measuring Tg with DSC: A Complete Protocol for Pharmaceutical Material Scientists
This article provides a comprehensive guide to Differential Scanning Calorimetry (DSC) for determining the glass transition temperature (Tg) of amorphous pharmaceutical solids.
Measuring Tg with DSC: A Complete Protocol for Pharmaceutical Material Scientists
Abstract
This article provides a comprehensive guide to Differential Scanning Calorimetry (DSC) for determining the glass transition temperature (Tg) of amorphous pharmaceutical solids. Aimed at researchers and formulation scientists, it covers the fundamental principles of the glass transition, step-by-step methodological protocols, troubleshooting for common issues, and validation strategies. The content addresses critical needs in drug development, from ensuring amorphous stability and predicting shelf-life to optimizing lyophilization cycles and understanding molecular mobility.
Understanding the Glass Transition: Why Tg is Critical for Drug Stability and Performance
The glass transition temperature (Tg) is a critical material property defining the reversible transition from a hard, amorphous glassy state to a soft, rubbery state. Within pharmaceutical and polymer science, precise Tg measurement is paramount for predicting product stability, solubility, and performance. This Application Note, framed within a broader thesis on Differential Scanning Calorimetry (DSC) protocol development, details the principles, current methodologies, and protocols for accurate Tg determination relevant to researchers and drug development professionals.
Fundamental Principles and Quantitative Data
The glass transition is a second-order endothermic shift, not a peak, characterized by a change in heat capacity (ΔCp). The measured Tg value is influenced by the material's thermal history, molecular weight, and plasticizer content (e.g., water).
Table 1: Representative Tg Values for Common Pharmaceutical Polymers and Amorphous Drugs
| Material | Tg (°C) Dry | Tg (°C) at 3% Moisture | ΔCp (J/g°C) | Reference Year |
|---|---|---|---|---|
| Polyvinylpyrrolidone (PVP K30) | 165 | ~80 | 0.47 | 2023 |
| Hydroxypropyl Methylcellulose (HPMC) | 155 | ~100 | 0.39 | 2023 |
| Amorphous Sucrose | 70 | -20 | 0.60 | 2024 |
| Indomethacin (amorphous) | 45 | N/A | 0.42 | 2024 |
| Poly(lactic-co-glycolic acid) (PLGA 50:50) | 45-50 | N/A | 0.35 | 2023 |
Table 2: Impact of Experimental Parameters on Measured Tg (DSC)
| Parameter | Typical Range | Effect on Measured Tg | Recommended Standard for Protocol |
|---|---|---|---|
| Heating Rate (°C/min) | 1 - 20 | Increases ~3°C per 10°C/min increase | 10 °C/min |
| Sample Mass (mg) | 3 - 10 | Larger mass can broaden transition | 5 - 8 mg |
| Hermetic Seal Integrity | Critical for hygroscopic samples | Moisture loss lowers Tg; leakage invalidates data | Crucible must be hermetically sealed |
| Data Analysis Method | Midpoint, Onset, Inflection | Midpoint most common; Onset ~2-5°C lower | Report method used (Midpoint ASTM E1356) |
Experimental Protocol: Standard DSC for Tg Measurement
This detailed protocol is designed for a TA Instruments Q2000 DSC or equivalent, within a controlled laboratory environment (23°C, <20% RH).
A. Materials and Reagent Solutions
- Research Reagent Solutions & Essential Materials:
- Hermetic Aluminum T-Crucibles with Lids: Provide a sealed environment to prevent moisture loss/uptake during analysis.
- Microbalance (0.001 mg accuracy): For precise sample weighing.
- Desiccator with P₂O₅: For dry storage of samples and crucibles.
- Liquid Nitrogen Cooling System (LNCS) or Intracooler: Enables sub-ambient temperature control for low-Tg materials.
- Indium Standard (Tm = 156.6°C, ΔHf = 28.5 J/g): For calibration of temperature and enthalpy.
- Empty Hermetic Crucible: Serves as the reference pan.
- Nitrogen Gas (High Purity, 50 mL/min flow): Inert purge gas to prevent oxidative degradation.
B. Procedure
- Instrument Calibration: Calibrate the DSC for temperature and enthalpy using pure Indium. Validate performance using a well-characterized standard (e.g., amorphous sucrose).
- Sample Preparation:
- Pre-dry the sample if necessary, under vacuum over P₂O₅ for 24h.
- Weigh 5.0 ± 0.5 mg of sample into a tared hermetic aluminum crucible.
- Seal the crucible immediately using the crucible press to ensure an airtight seal.
- Prepare an identically sealed empty crucible as the reference.
- Experimental Parameters Setup:
- Purge Gas: Nitrogen at 50 mL/min.
- Temperature Program:
- Equilibration: -20°C (or 50°C below expected Tg).
- Isothermal Hold: 5 min.
- Heating Scan: From equilibration temperature to 50°C above expected Tg at 10°C/min.
- Cooling Scan (Optional, for erasing thermal history): Cool back to starting temperature at 20°C/min.
- Second Heating Scan: Repeat the heating scan at 10°C/min. Analyze the Tg from this second heating scan.
- Data Acquisition: Load the sample and reference. Initiate the method. Visually inspect the seal post-run.
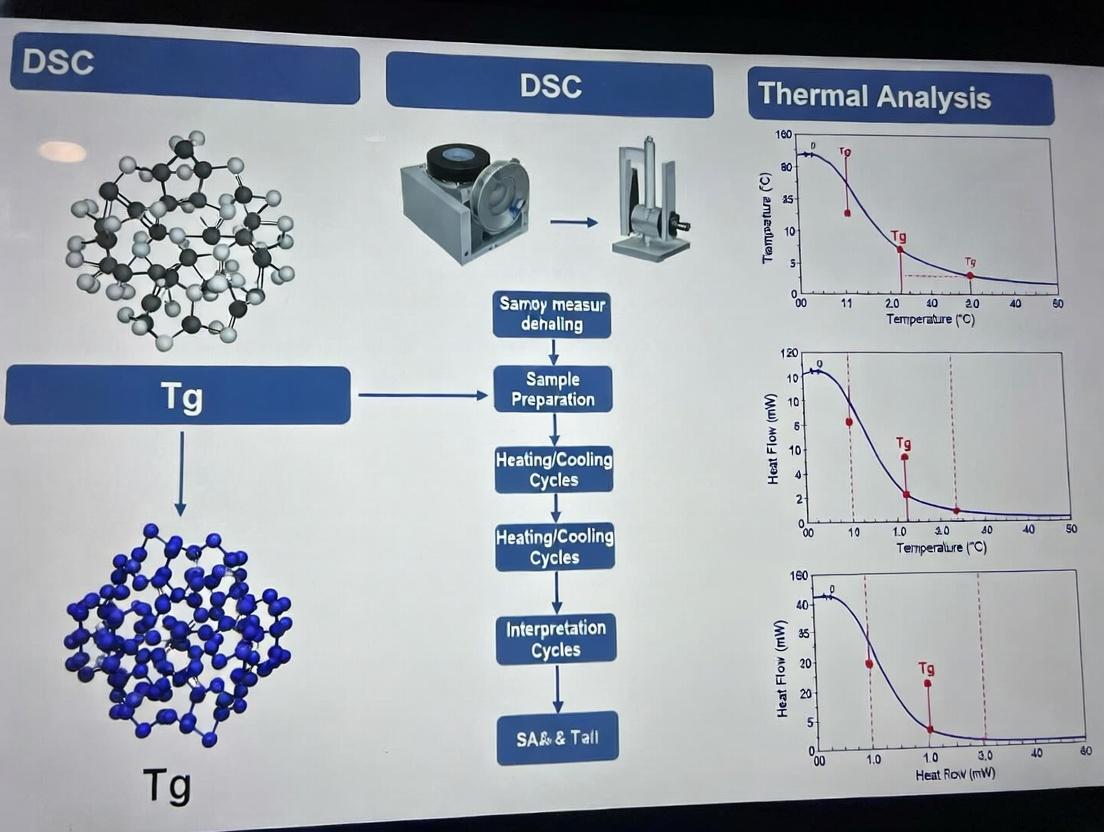
- Data Analysis (According to ASTM E1356):
- Plot heat flow (W/g) vs. Temperature (°C).
- Draw tangents to the baselines before and after the transition step.
- The glass transition temperature (Tg) is reported as the midpoint—the temperature at which the heat flow curve is equidistant between the two extrapolated baselines.
- Report the onset and endset temperatures as supplementary data.
- Calculate and report the change in heat capacity (ΔCp).
Workflow and Data Interpretation Diagrams
Diagram 1: DSC Tg Measurement Protocol Workflow
Diagram 2: Tg Determination from DSC Heat Flow Curve
Application Notes: The Critical Role of Tg
The glass transition temperature (Tg) is a fundamental property of amorphous pharmaceutical solids, dictating their physical stability, dissolution behavior, and processability. Within the context of a broader thesis on Differential Scanning Calorimetry (DSC) protocols for measuring Tg, its accurate determination is paramount for rational formulation design.
Stability: Below Tg, molecules in an amorphous solid (e.g., an amorphous solid dispersion) are frozen in a high-viscosity, glassy state, leading to kinetic stabilization of the drug. At storage temperatures (T) > Tg, molecular mobility increases exponentially, leading to physical instabilities like crystallization, which can compromise bioavailability. The rule of thumb is to store at least 50°C below Tg for long-term stability.
Solubility & Dissolution: Amorphous forms, stabilized below their Tg, offer enhanced apparent solubility and dissolution rates compared to their crystalline counterparts—a key strategy for bioavailability enhancement of poorly soluble drugs (BCS Class II/IV). The dissolution performance is directly influenced by the physical state stability governed by Tg.
Processing: Common pharmaceutical processes like hot-melt extrusion (HME), spray drying, and freeze-drying involve the transformation of materials through or into the amorphous state. Tg determines process temperatures (e.g., extrusion temperature must be above Tg for flow) and influences residual stresses and stability post-processing.
Quantitative Data Summary:
Table 1: Glass Transition Temperatures of Common Pharmaceutical Polymers and Their Role
| Material | Approximate Tg (°C) | Pharmaceutical Role & Implication |
|---|---|---|
| Polyvinylpyrrolidone (PVP K30) | ~170-180 | High Tg carrier; increases system Tg, stabilizing drug. |
| Hydroxypropyl Methylcellulose Acetate Succinate (HPMCAS) | ~120-135 | pH-dependent polymer for enteric solid dispersions. |
| Soluplus (PVA-PEG graft copolymer) | ~70 | Low Tg polymer aids processability in HME. |
| Copolovidone (PVP-VA64) | ~105-110 | Common spray-drying carrier, balances Tg & processability. |
| Sucrose | ~70-75 | Lyoprotectant in freeze-drying; Tg critical for cake stability. |
| Indomethacin (model drug) | ~45-50 | Low Tg drug requires high-Tg polymer for stabilization. |
| Itraconazole (model drug) | ~60 | Poorly soluble drug often formulated in solid dispersions. |
Table 2: Stability Rules of Thumb Based on Tg (T = Storage Temperature)*
| Condition | Empirical Rule | Practical Implication |
|---|---|---|
| Long-term Stability | T < Tg - 50°C | Ideal for commercial shelf life. |
| Short-term / Accelerated | T < Tg - 20°C | May be acceptable for early development. |
| Risk of Crystallization | T > Tg | Significant molecular mobility; rapid degradation of amorphous advantage. |
Experimental Protocols
Protocol 1: Standard DSC Protocol for Tg Determination of an Amorphous Solid Dispersion
Context: Core methodology for thesis research.
Objective: To determine the glass transition temperature of a spray-dried amorphous solid dispersion using DSC. Materials: DSC instrument (e.g., TA Instruments Q2000), nitrogen purge gas, Tzero aluminum pans and lids, analytical balance, amorphous solid dispersion sample, inert reference (empty pan).
Procedure:
- Calibration: Calibrate the DSC for temperature and enthalpy using indium and zinc standards.
- Sample Preparation: Weigh 5-10 mg of the solid dispersion powder precisely into a Tzero aluminum pan. Hermetically seal the pan with a lid using a press.
- Experimental Setup: Load the sealed sample pan and an empty reference pan into the DSC. Set a nitrogen purge flow rate of 50 mL/min.
- Thermal Method: a. Equilibrate at 0°C. b. Ramp temperature from 0°C to 200°C at a scan rate of 10°C/min. c. Isothermal for 5 minutes at 200°C to erase thermal history. d. Cool rapidly to 0°C at 50°C/min. e. Re-equilibrate at 0°C. f. Perform the measurement scan: heat from 0°C to 200°C at 10°C/min.
- Data Analysis: In the software, plot heat flow (W/g) vs. temperature. Identify the Tg as the midpoint of the step-change in heat flow (inflection point) on the second heating scan. Report the onset, midpoint, and endpoint temperatures.
Protocol 2: Determination of Plasticization Effect by Moisture
Objective: To assess the depression of Tg caused by moisture absorption, a critical stability factor. Materials: As in Protocol 1, plus a desiccator and saturated salt solutions for humidity conditioning (e.g., 75% RH using NaCl slurry).
Procedure:
- Condition separate samples of the amorphous solid dispersion at controlled relative humidities (0% RH, 33% RH, 75% RH) for 7 days in desiccators at 25°C.
- Quickly weigh and seal each conditioned sample in a DSC pan.
- Analyze each sample using the thermal method from Protocol 1 (steps 4f).
- Plot the measured Tg versus %RH or moisture content. Use the Gordon-Taylor equation to model the relationship and predict Tg at various moisture levels.
Diagrams
The Scientist's Toolkit: Key Research Reagent Solutions & Materials
Table 3: Essential Materials for Tg-Focused Pharmaceutical Research
| Item | Function / Role |
|---|---|
| Differential Scanning Calorimeter (DSC) | Primary instrument for direct measurement of Tg via heat flow change. |
| Hermetic Tzero Aluminum Pans & Lids | Ensures sealed environment, preventing sample volatilization/oxidation during heating. |
| High-Purity Nitrogen Gas | Inert purge gas to prevent oxidative degradation during DSC analysis. |
| Calibration Standards (Indium, Zinc) | For accurate temperature and enthalpy calibration of the DSC. |
| Model Amorphous Drugs (e.g., Indomethacin) | Low-Tg drugs used in method development and stability studies. |
| Pharmaceutical Polymers (PVP, HPMCAS, Copovidone) | Carriers to formulate solid dispersions; their Tg modulates system properties. |
| Humidity Control Chambers/Saturated Salt Solutions | For conditioning samples at specific RH to study moisture plasticization effects. |
| Microbalance (0.01 mg accuracy) | Precise sample weighing for reproducible DSC results. |
| Lyophilizer/Spray Dryer | Equipment to manufacture amorphous materials for study. |
| Hot-Melt Extruder (Lab-scale) | For processing amorphous solid dispersions; Tg guides extrusion temperature. |
Within the broader thesis on establishing robust Differential Scanning Calorimetry (DSC) protocols for measuring the glass transition temperature (Tg) of amorphous solid dispersions and other pharmaceutical systems, this application note focuses on the critical link between Tg, molecular mobility, and chemical degradation kinetics. The central thesis posits that accurately determining Tg via optimized DSC protocols is not an endpoint but a vital starting point for predicting and mitigating instability. The physical state below and above Tg governs molecular mobility, which is the principal driver of degradation pathways like hydrolysis, oxidation, and deamidation in solid-state formulations.
Core Scientific Principles: Linking Tg, Molecular Mobility, and Degradation
The relationship between temperature, molecular mobility, and reaction rates is described by the Williams-Landel-Ferry (WLF) and Arrhenius equations. Near and above Tg, molecular mobility increases dramatically.
Quantitative Framework: The rate of a solid-state chemical degradation reaction (k) is related to molecular mobility and temperature. Above Tg, the temperature dependence often follows the WLF equation: log( aT ) = -C1 (T - Tref) / (C2 + T - Tref) where aT is the mobility shift factor, T is temperature, Tref is a reference temperature (often Tg), and C1 and C2 are system-specific constants. Below Tg, in the glassy state, the temperature dependence may follow an Arrhenius relationship but with a significantly higher activation energy due to mobility restrictions.
Table 1: Representative Tg Values and Associated Degradation Rate Constants for Model Compounds
| Compound/Formulation | Tg (°C) | Storage T (°C) | T - Tg | Degradation Rate Constant (k, day⁻¹) | Primary Degradation Mode |
|---|---|---|---|---|---|
| Amorphous Sucrose | 70 | 25 | -45 | 0.0005 | Hydrolysis |
| Amorphous Sucrose | 70 | 60 | -10 | 0.002 | Hydrolysis |
| Amorphous Sucrose | 70 | 75 | +5 | 0.015 | Hydrolysis & Crystallization |
| Indomethacin ASD (PVP) | 105 | 40 (Dry) | -65 | <0.0001 | Oxidation |
| Indomethacin ASD (PVP) | 105 | 40 (75% RH) | -65* | 0.0012 | Hydrolysis |
| Lyophilized mAb Formulation | 110 | 25 | -85 | 0.00005 | Deamidation |
RH plasticizes the system, effectively lowering the Tg.
Table 2: Effect of Stabilizers/Plasticizers on Tg and Degradation Half-life (t90)
| API | Polymer/Excipient | Tg of Blend (°C) | ΔTg from Pure API | t90 at 40°C/75% RH (Months) |
|---|---|---|---|---|
| Ritonavir | None (amorphous) | 50 | - | 1 |
| Ritonavir | PVP-VA64 | 105 | +55 | 24 |
| Celecoxib | None (amorphous) | 55 | - | 3 |
| Celecoxib | HPMCAS-LF | 120 | +65 | >36 |
| Felodipine | PEO (plasticizer) | 35 | -15 | 6 |
Experimental Protocols
Protocol 4.1: DSC Protocol for Tg Determination in Stability Context
Objective: To accurately measure the Tg of a formulation for subsequent mobility/degradation modeling.
- Sample Preparation: Pre-dry sample if hygroscopic. Precisely weigh 5-10 mg into a hermetically sealed DSC pan with a pinhole lid to allow moisture escape during initial heating, if necessary for mimicking storage conditions.
- Instrument Calibration: Calibrate DSC for temperature and enthalpy using indium and zinc standards.
- Method Parameters:
- Purge Gas: Dry nitrogen at 50 mL/min.
- Heating Rate: Use a standard rate of 10°C/min. For complex systems, a second scan after quenching may be required to remove thermal history.
- Temperature Range: Typically -20°C to 150°C or 20°C above expected degradation onset.
- Cycling (Optional): For complex dispersions, use a heat-cool-reheat cycle to identify reversible Tg.
- Data Analysis: Tg is taken as the midpoint of the step change in heat capacity. Report onset, midpoint, and endpoint temperatures. Note any enthalpic recovery peaks or crystallization events.
Protocol 4.2: Isothermal Stability Study Correlated to Tg
Objective: To empirically determine degradation kinetics at temperatures relative to Tg.
- Condition Selection: Prepare aliquots of the amorphous solid dispersion.
- Storage Conditions: Store samples in controlled stability chambers at temperatures strategically chosen relative to the measured Tg (e.g., Tg - 50°C, Tg - 30°C, Tg - 10°C, Tg + 5°C). Maintain constant relative humidity (e.g., 0%, 32%, 75% RH).
- Sampling: Remove triplicate samples at predetermined time points (e.g., 0, 1, 2, 4, 8, 12 weeks).
- Analysis: Assay for:
- Chemical Purity: HPLC for API degradation products.
- Physical State: XRD to check for crystallization; DSC to monitor any Tg changes.
- Moisture Content: Karl Fischer titration.
- Kinetic Modeling: Plot degradation (e.g., % potency remaining) vs. time. Determine rate constants (k) at each storage T. Construct an Arrhenius or WLF plot using (T - Tg) as the driving force.
The Scientist's Toolkit: Key Reagent Solutions & Materials
| Item | Function & Relevance to Tg/Degradation Studies |
|---|---|
| Hermetic DSC Pans with Pinhole Lids | Allows for controlled moisture escape during heating, crucial for measuring Tg under conditions relevant to humid storage. |
| Standard Reference Materials (Indium, Zinc) | Essential for accurate temperature and enthalpy calibration of the DSC, ensuring Tg measurement precision. |
| Desiccants (e.g., P2O5, molecular sieves) | Used to create dry storage conditions (0% RH) to isolate temperature effects from plasticization. |
| Saturated Salt Solutions (e.g., MgCl2, NaCl, KNO3) | Used in desiccators to generate precise, constant relative humidity environments (32%, 75%, 94% RH) for stability studies. |
| High-Performance Polymers (PVP, HPMCAS, PVP-VA) | Common polymeric stabilizers used in amorphous solid dispersions to elevate Tg and inhibit molecular mobility. |
| Plasticizers (e.g., Glycerol, PEG 400) | Used to systematically lower Tg in model studies to probe the Tg-degradation relationship. |
Visualizations
Diagram 1: The Tg-Mobility-Degradation Cascade
Diagram 2: DSC Protocol Workflow for Stability Assessment
Application Notes
Amorphous solid dispersions (ASDs) and lyophilized formulations are critical for enhancing the bioavailability of poorly water-soluble active pharmaceutical ingredients (APIs). Differential Scanning Calorimetry (DSC) is the primary technique for characterizing the glass transition temperature (Tg), a key parameter dictating the physical stability of these amorphous systems. The Tg of an amorphous API-polymer blend is not a fixed value but is influenced by factors such as moisture content, thermal history, and the specific interactions between components. Recent research emphasizes the role of polymer chemistry (e.g., vinylpyrrolidone-based vs. acrylate-based) in modulating Tg and inhibiting crystallization. For lyophilized products, the Tg of the maximally freeze-concentrated solute (Tg') is a vital parameter for optimizing the freeze-drying cycle and ensuring cake stability. The following tables summarize key quantitative relationships.
Table 1: Representative Tg Values for Common Amorphous Systems
| Material / System | Typical Tg Range (°C) | Critical Influencing Factor | Reference Year |
|---|---|---|---|
| Amorphous Indomethacin | 42 - 49 | Polymorphic form, heating rate | 2023 |
| PVP-VA 64 (Polymer) | 106 - 108 | Molecular weight, moisture | 2024 |
| Indomethacin: PVP-VA 64 (70:30) | ~85 - 90 | Drug loading, hydrogen bonding | 2023 |
| HPMCAS (Polymer) | 120 - 135 | Acyl substitution level | 2024 |
| Lyophilized Sucrose Cake | -32 to -34 (Tg') | Residual moisture, bulking agent | 2024 |
| Lyophilized mAb Formulation (with trehalose) | 75 - 85 (Tg dry) | Protein: sugar ratio, annealing step | 2023 |
Table 2: Impact of Moisture on Tg of Selected Amorphous Formulations
| Formulation | Tg (Dry) (°C) | Tg at 3% RH (°C) | Tg at 50% RH (°C) | ΔTg/ΔMoisture |
|---|---|---|---|---|
| Amorphous Sorafenib | 79.5 | 65.2 | 32.1 | High |
| Itraconazole: HPMC 3:7 | 94.3 | 87.6 | 45.8 | High |
| Spray-Dried Manitol | 15.0 | N/A | -10.0 | Very High |
| Freeze-Dried Ritonavir Solid Dispersion | 88.0 | 82.5 | 70.1 | Moderate |
Experimental Protocols
Protocol 1: Standard DSC Protocol for Measuring Tg in Amorphous Solid Dispersions
Objective: To determine the glass transition temperature (Tg) of an amorphous API-polymer blend. Materials: DSC instrument (e.g., TA Instruments Q2000, Mettler Toledo DSC3), hermetic Tzero pans and lids, analytical balance, dry nitrogen purge gas. Procedure:
- Sample Preparation: Precisely weigh 5-10 mg of the ASD powder using an analytical balance.
- Pan Sealing: Transfer the sample to a hermetic Tzero pan and seal it with a lid using the sample press. Prepare an empty sealed pan as a reference.
- Instrument Calibration: Calibrate the DSC for temperature and enthalpy using indium and zinc standards.
- Method Programming: Create a method with the following segments: a. Equilibrate at 0°C. b. Isothermal hold for 5 min. c. Ramp from 0°C to 200°C at a heating rate of 10°C/min. d. Isothermal hold for 5 min to erase thermal history. e. Cool rapidly to 0°C at 50°C/min. f. Ramp from 0°C to 200°C at 10°C/min (this second heating scan is used for analysis).
- Data Analysis: In the analysis software, plot heat flow (W/g) vs. Temperature. Identify the Tg as the midpoint of the step-change in heat flow (inflection point) on the second heating scan. Report the onset, midpoint, and endpoint temperatures.
Protocol 2: Determination of Tg' for Lyophilized Formulation Development
Objective: To measure the glass transition temperature of the maximally freeze-concentrated solute (Tg') for freeze-drying cycle development. Materials: DSC with liquid nitrogen cooling system, hermetic high-volume pans (e.g., 40µL), solution of the formulation (API + excipients in water). Procedure:
- Sample Loading: Pipette 10-20 µL of the aqueous formulation solution directly into a hermetic DSC pan. Seal the pan immediately.
- Method Programming: a. Equilibrate at 25°C. b. Cool to -70°C at a rapid rate (e.g., 20°C/min) to fully vitrify the sample. c. Isothermal hold for 5 min. d. Heat to 25°C at a controlled rate (5°C/min).
- Data Analysis: Analyze the first heating scan. Identify the Tg' as the midpoint of the glass transition step observed in the temperature range typically between -50°C and -30°C for many sugar-based formulations. This event is often followed by a devitrification exotherm (ice crystallization) and an endothermic melt.
Protocol 3: Modulated DSC (MDSC) for Complex Transitions
Objective: To separate reversing (heat capacity-related, e.g., Tg) from non-reversing (kinetic, e.g., relaxation, crystallization) thermal events. Materials: As per Protocol 1. Procedure:
- Follow steps 1-3 from Protocol 1.
- Method Programming: Use a standard heat-only MDSC method. a. Equilibrate at 0°C. b. Heat from 0°C to 200°C at an underlying heating rate of 2°C/min, with a modulation amplitude of ±0.5°C every 60 seconds.
- Data Analysis: Process the data to generate Reversing Heat Flow and Non-Reversing Heat Flow signals. The Tg is identified as a step change in the Reversing Heat Flow signal. The Non-Reversing signal may show enthalpy relaxation exotherms near the Tg.
Visualizations
Diagram Title: Factors Influencing Amorphous Solid Dispersion Physical Stability
Diagram Title: DSC Glass Transition Measurement Protocol Steps
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function & Relevance |
|---|---|
| Hermetic Tzero Pans & Lids | Sealed aluminum pans that prevent sample loss/contamination and control atmosphere (e.g., for moisture-sensitive samples). Essential for accurate Tg measurement. |
| Standard Reference Materials (Indium, Zinc) | High-purity metals with known melting points and enthalpies for precise temperature and heat flow calibration of the DSC instrument. |
| Dry Nitrogen Purge Gas | Inert gas supply to maintain a dry, oxide-free atmosphere in the DSC cell, preventing sample degradation and ensuring a stable baseline. |
| PVP-VA 64 (Copovidone) | A widely used polymeric carrier in ASDs. Its Tg (~106°C) and hydrogen-bonding capability stabilize amorphous APIs. Serves as a model polymer for method development. |
| Amorphous Indomethacin | A model poorly water-soluble API that readily forms a stable glass. Frequently used as a benchmark material for validating Tg measurement protocols. |
| Trehalose Dihydrate (Lyophilization Grade) | A non-reducing disaccharide used as a cryoprotectant and lyoprotectant. Critical for measuring Tg' in biopharmaceutical lyophilization development. |
| HPMCAS (Hypromellose Acetate Succinate) | An enteric polymer used in pH-dependent ASD release. Its high Tg (120-135°C) makes it a good test material for high-temperature transitions. |
| Modulated DSC (MDSC) Software License | Enables the deconvolution of complex thermal events, separating the reversible Tg from overlapping enthalpic relaxation, crucial for complex formulations. |
Differential Scanning Calorimetry (DSC) is a fundamental thermoanalytical technique for measuring the heat flow into or out of a sample as a function of temperature or time. Within pharmaceutical and materials research, particularly in the study of amorphous solid dispersions and polymer-based drug delivery systems, the precise measurement of the glass transition temperature (Tg) via DSC is critical for understanding physical stability, miscibility, and performance.
Core Principles of Heat Flow Measurement
DSC operates by comparing the heat flow required to increase the temperature of a sample and an inert reference. The primary measurement modes are:
- Heat-Flux DSC: Sample and reference are connected by a low-resistance heat flow path. The temperature difference (ΔT) between them, resulting from thermal events, is proportional to the heat flow difference.
- Power-Compensation DSC: Separate furnaces and sensors for the sample and reference. The instrument actively supplies differential power to maintain both at the same temperature, with this power being the direct measured signal.
The fundamental relationship is described by: dq/dt = f(T or t), where dq/dt is the heat flow (typically mW or mJ/s), and the independent variable is temperature (T) or time (t). For Tg determination, the observed baseline shift corresponds to a change in the sample's heat capacity (Cp).
Quantitative Data on Key Thermal Transitions
| Thermal Transition | Typical Sign in DSC Curve (Heat Flow) | Primary Information Obtained | Common in Pharmaceuticals |
|---|---|---|---|
| Glass Transition (Tg) | Endothermic Step Shift | Change in heat capacity (ΔCp), midpoint Tg | Amorphous APIs, polymers, solid dispersions |
| Melting (Tm) | Sharp Endothermic Peak | Melting temperature, enthalpy (ΔH), purity | Crystalline APIs, excipients |
| Crystallization | Sharp Exothermic Peak | Crystallization temperature & enthalpy | Stability of amorphous forms |
| Decomposition | Broad Endo/Exothermic Peak | Onset temperature, degradation profile | Excipient compatibility, stability |
Experimental Protocols for Tg Measurement
Protocol 1: Standard Tg Determination for a Polymer or Amorphous API
Objective: To accurately determine the midpoint glass transition temperature (Tg) of an amorphous drug substance.
Materials & Equipment:
- Differential Scanning Calorimeter (e.g., TA Instruments Q系列, Mettler Toledo DSC 3)
- Hermetically sealed Tzero aluminum pans and lids
- Analytical balance (accuracy ±0.01 mg)
- Sample: 5-10 mg of amorphous material
- Dry Nitrogen purge gas (50 mL/min flow rate)
Procedure:
- Preparation: Precisely weigh 5-10 mg of sample into a tared Tzero aluminum pan. Crimp the lid using a hermetic sealer to ensure an airtight seal. Prepare an empty, sealed pan as the reference.
- Instrument Calibration: Calibrate the DSC for temperature and enthalpy using indium (Tm = 156.6°C, ΔH = 28.4 J/g).
- Method Programming: Load the following temperature program:
- Equilibration: -20°C
- Isotherm: Hold for 5 min.
- Ramp: Heat from -20°C to 150°C at a rate of 10°C/min.
- Data Acquisition: Place the sample and reference pans in the furnace. Start the method under a nitrogen purge.
- Data Analysis: In the analysis software, plot heat flow (W/g) vs. temperature. Identify the glass transition region. Perform a tangent fit to determine the onset, midpoint, and endpoint temperatures. The midpoint Tg is reported.
Protocol 2: Tg Measurement for a Lyophilized Protein Formulation
Objective: To assess the Tg' (glass transition of the maximally freeze-concentrated solution) and Tg of the dry lyophile, critical for process and storage stability.
Materials & Equipment:
- Power-compensation DSC with intracooler/subambient accessory.
- Hermetic stainless steel high-volume pans or Tzero pans.
- Pre-lyophilized cake or liquid formulation for pan loading.
Procedure:
- Sample Loading (for Tg'): For a liquid formulation, load 10-20 µL into a pan and seal non-hermetically to allow for vapor pressure.
- Method for Tg':
- Equilibrate at 25°C.
- Cool to -60°C at 10°C/min.
- Isotherm for 5 min.
- Heat to 25°C at 5°C/min. Analyze the reversible thermal event (change in heat capacity) as Tg'.
- Method for Dry Tg: For the lyophilized cake, follow Protocol 1, using a hermetically sealed pan and a heating ramp from -20°C to 150°C at 10°C/min.
Visualization of DSC Workflow and Data Interpretation
DSC Protocol Workflow for Tg
Analyzing Tg on a DSC Thermogram
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item | Function & Importance in DSC for Tg |
|---|---|
| Hermetic Sealed Pans (Tzero/Aluminum) | Prevents solvent loss during heating, crucial for accurate Tg measurement of hydrated samples or materials with residual solvent. |
| Nitrogen Purge Gas (High Purity, >99.9%) | Inert atmosphere that prevents oxidative degradation of samples during heating and ensures stable thermal baseline. |
| Calibration Standards (Indium, Zinc) | Certified reference materials for accurate temperature and enthalpy calibration of the DSC cell, mandatory for valid data. |
| Desiccants (e.g., Silica Gel) | For dry storage of DSC pans and samples; moisture sensitive amorphous materials must be kept dry prior to analysis. |
| Sample Encapsulation Press | Tool for crimping hermetic pans, ensuring a uniform and leak-proof seal critical for reliable measurements. |
| Microbalance (0.01 mg resolution) | Accurate sample weighing (typically 3-10 mg) is essential for precise normalization of heat flow data (W/g). |
| Liquid Nitrogen Cooling System | Provides sub-ambient temperature control for measuring Tg' of frozen systems or low-Tg polymers. |
Step-by-Step DSC Protocol for Accurate Tg Measurement in Pharmaceuticals
1. Introduction: Context within DSC-Tg Research The precise determination of the Glass Transition Temperature (Tg) via Differential Scanning Calorimetry (DSC) is critical in pharmaceutical development for characterizing amorphous solid dispersions, biologics, and polymer-based formulations. The validity of Tg data is inherently dependent on rigorous pre-test sample preparation. Uncontrolled sample history (e.g., processing, storage humidity), inadequate conditioning, and imperfect hermetic sealing can lead to significant artifacts, including enthalpy relaxation peaks, plasticization-induced Tg depression, or sample decomposition, thereby compromising the research thesis.
2. Application Notes & Protocols
2.1. Documenting and Controlling Sample History
- Protocol: Establish a standardized Sample History Data Sheet for every DSC specimen.
- Methodology:
- Record the synthesis or processing method (e.g., spray drying, melt quenching, lyophilization), including cooling/heating rates.
- Document storage conditions: time, temperature, and relative humidity (RH) from production to analysis.
- Note any prior thermal or mechanical treatment.
- For comparative studies, ensure all samples in a series share an identical thermal history prior to the designated DSC protocol.
2.2. Sample Conditioning Protocol
- Objective: To equilibrate the sample to a known, reproducible state of moisture content and enthalpy, minimizing relaxation phenomena during the first DSC scan.
- Detailed Protocol:
- Desiccation (for moisture-sensitive samples): Place the bulk material in a desiccator over phosphorus pentoxide (P₂O₅) or a similar anhydrous desiccant at room temperature for a minimum of 48 hours.
- Humidity Conditioning (for controlled hydration): Use a controlled humidity chamber or saturated salt solutions to condition samples at a specific RH (e.g., 0%, 32%, 75% RH) at constant temperature (e.g., 25°C) for ≥7 days to ensure equilibrium.
- Thermal Annealing (to relieve stresses): For studies targeting the "equilibrated" glassy state, anneal the sample in a sealed pan at a temperature 5-10°C below its expected Tg for a defined period (e.g., 2-24 hours), followed by quench-cooling.
2.3. Hermetic Seal Preparation Protocol
- Objective: To prevent mass loss (water/solvent volatilization) and avoid oxidative degradation during the DSC run, ensuring the measured transition is intrinsic to the material.
- Detailed Protocol:
- Material Preparation: Use high-purity aluminum hermetic pans and lids. Handle with clean tweezers.
- Loading: Precisely weigh (typically 3-10 mg) the conditioned sample into the pan using a microbalance.
- Sealing: Place the lid on the pan and seal using a laboratory hermetic press. Apply consistent, firm pressure.
- Verification: Visually inspect the seal under magnification for uniformity and absence of crimping flaws. Weigh the sealed pan to confirm no mass loss occurred during sealing. A post-run mass check is also recommended.
3. Quantitative Data Summary
Table 1: Impact of Moisture Conditioning on Measured Tg of a Model Polymer (e.g., PVP)
| Conditioning RH (%) | Equilibrium Moisture Content (wt%)* | Tg Onset (°C) | Tg Midpoint (°C) | Observation |
|---|---|---|---|---|
| 0 (P₂O₅) | 0.1 | 175.5 | 177.2 | Baseline |
| 32 | 3.5 | 155.1 | 157.8 | Tg depression |
| 75 | 8.2 | 121.7 | 124.5 | Significant Tg depression & broadening |
*Values are illustrative. Actual data must be experimentally determined.
Table 2: Effect of Hermetic Seal Integrity on DSC Measurement Artifacts
| Seal Condition | Observation in DSC Thermogram | Consequence for Tg Analysis |
|---|---|---|
| Proper Hermetic | Clean baseline, distinct Tg step. | Accurate, reproducible Tg value. |
| Non-Hermetic | Endothermic drift or peak preceding/overlapping Tg due to solvent evaporation. | Tg onset obscured, inaccurate, variable. |
| Imperfect/Leaky | Broad, shifting Tg step; may show exothermic decomposition events at higher temperatures. | Unreliable data; risk of misinterpretation. |
4. Visualizations
Title: DSC Tg Analysis Pre-Test Workflow (6 steps)
Title: Impact of Poor Prep on DSC-Tg Data & Research
5. The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for DSC-Tg Pre-Test Preparation
| Item/Reagent | Function & Importance |
|---|---|
| Hermetic DSC Pans & Lids | High-purity aluminum pans ensure inert, pressure-tight encapsulation to prevent mass transfer. Crucial for volatile samples. |
| Hermetic Sealing Press | Provides uniform, leak-free crimping of pans. Manual or hydraulic presses are standard. |
| Microbalance (≥0.01 mg) | Enables precise sample weighing (3-10 mg range) for quantitative heat flow measurement. |
| Desiccator & P₂O₅ | Provides an anhydrous environment (<1% RH) for drying moisture-sensitive samples. |
| Saturated Salt Solutions | Creates defined humidity environments (e.g., LiCl, MgCl₂, NaCl salts) for controlled conditioning. |
| Humidity/Temp. Chamber | Allows precise, programmable conditioning of samples at specific T/RH for kinetic studies. |
| Dry Box/Glovebox | For handling extremely hygroscopic or oxygen-sensitive materials during pan loading. |
| Calibrated Reference Materials | Indium, Zinc for temperature/enthalpy calibration of the DSC, ensuring instrument validity. |
Instrument Calibration and Selection of Crucible Type (e.g., Tzero, Standard Aluminum)
Within the broader thesis on establishing robust DSC protocols for measuring the glass transition temperature (Tg) of amorphous solid dispersions in pharmaceutical research, the calibration of the instrument and the selection of appropriate crucibles are foundational steps. These factors critically influence the accuracy, precision, and reproducibility of Tg measurements, which are essential for predicting drug product stability and performance. This application note details standardized protocols and data-driven selection criteria.
Instrument Calibration: Protocols and Data
Proper calibration ensures the DSC signal accurately reflects thermal events. A multi-point calibration using certified reference materials is recommended.
Table 1: Key Calibration Standards for Tg Research
| Standard Material | Certified Melting Point (°C) | Primary Use in Calibration | Notes for Tg Applications |
|---|---|---|---|
| Indium (In) | 156.60 ± 0.10 | Temperature & Enthalpy | Primary low-T range standard. |
| Tin (Sn) | 231.93 ± 0.10 | Temperature | Mid-range verification. |
| Zinc (Zn) | 419.53 ± 0.10 | Temperature | High-range verification. |
| Cyclohexane | -87.06 | Temperature | Sub-ambient calibration. |
| Sapphire (Al₂O₃) | N/A (Heat Capacity Std.) | Heat Capacity | Critical for Cp baseline for Tg. |
Protocol 1.1: Temperature and Enthalpy Calibration
- Equipment Prep: Ensure the DSC cell is clean and purged with nitrogen (50 mL/min).
- Crucible Selection: Use hermetic Tzero pans for Indium to prevent oxidation. Crimp standard aluminum pans for In, Sn, Zn.
- Sample Prep: Precisely weigh 3-5 mg of standard (e.g., Indium) using a microbalance.
- Experimental Parameters:
- Heating Rate: 10 °C/min (matching intended Tg measurement rate).
- Temperature Range: 30 °C below to 30 °C above the standard's melt.
- Data Acquisition Rate: ≥1 Hz.
- Run: Perform triplicate runs of each standard.
- Analysis: In the DSC software, calibrate using the onset temperature for temperature calibration and the integrated peak area (ΔH) for enthalpy calibration.
Protocol 1.2: Heat Capacity (Cp) Calibration using Sapphire
- Three-Run Sequence:
- Run 1: Empty reference and sample pans (baseline).
- Run 2: Sapphire disk (15-25 mg) in sample pan.
- Run 3: Identical parameters for the drug/polymer sample.
- Parameters: Use the same heating rate and temperature range as for sample analysis.
- Software Calculation: The instrument software uses the three runs to calculate the sample's specific heat capacity, vital for accurate Tg step-height measurement.
Title: DSC Calibration Workflow for Tg Analysis
Crucible Selection: Impact on Tg Measurement
The crucible (pan) type influences sample environment, pressure, and heat transfer, affecting the observed Tg.
Table 2: Crucible Types for Pharmaceutical Tg Research
| Crucible Type | Seal Type | Pressure Tolerance | Key Advantages for Tg | Key Limitations |
|---|---|---|---|---|
| Standard Aluminum (Al) | Crimped | ~3 atm (Limited) | Excellent heat transfer, inexpensive, low thermal mass. | Volatiles can escape, not hermetic, susceptible to pan deformation. |
| Hermetic Aluminum (Al) | Volumetric (Cold-welded) | High (>10 atm) | Contains volatiles/solvents, prevents oxidation. | Requires specialized sealer, risk of over-pressure rupture. |
| Tzero Aluminum | Hermetic (Cold-welded) | High | Matched mass with lid for superior baseline, contains volatiles. | Higher cost, requires Tzero lids and sealer. |
| Tzero Hermetic | Hermetic (Cold-welded) | Very High | Best for volatile samples, superior baseline stability. | Highest cost, specialized tooling required. |
Protocol 2.1: Method for Evaluating Crucible Type on Measured Tg
- Sample Preparation: Prepare a uniform batch of an amorphous drug-polymer dispersion (e.g., 20% w/w API in PVPVA).
- Panning: Precisely weigh 5-10 mg samples into:
- a) Standard Al pan, crimped.
- b) Hermetic Al pan, sealed.
- c) Tzero Al pan with Tzero lid, sealed.
- DSC Parameters:
- Calibration: Perform fresh calibration per Protocol 1.1.
- Method: Heat from 25°C to 150°C at 10 °C/min.
- Purge: N₂ at 50 mL/min.
- Analysis: Measure Tg using the midpoint (half-step) method. Record onset, midpoint, endpoint, and ΔCp.
Table 3: Hypothetical Tg Data for Different Crucible Types (Model System)
| Crucible Type | Tg Onset (°C) | Tg Midpoint (°C) | ΔCp (J/g·°C) | Notes on Baseline Stability |
|---|---|---|---|---|
| Standard Al (Crimped) | 48.2 ± 0.5 | 51.5 ± 0.6 | 0.32 ± 0.02 | Baseline shift pre-Tg due to slight moisture loss. |
| Hermetic Al (Sealed) | 49.8 ± 0.3 | 52.9 ± 0.3 | 0.35 ± 0.01 | Sharper transition, contains residual solvent. |
| Tzero Al (Sealed) | 49.9 ± 0.2 | 53.0 ± 0.2 | 0.36 ± 0.01 | Smoothest, most stable baseline; highest precision. |
Title: Decision Tree for DSC Crucible Selection
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for DSC Tg Protocol Development
| Item | Function & Relevance to Tg Research |
|---|---|
| Certified Indium Standard | Primary calibrant for temperature and enthalpy; ensures reported Tg values are traceable to standards. |
| Sapphire Disk (Al₂O₃) | Reference material for specific heat capacity (Cp) calibration; required for accurate quantification of the ΔCp at Tg. |
| High-Purity Nitrogen Gas | Inert purge gas to prevent oxidation of samples and pans, ensuring stable, non-reactive atmosphere. |
| Tzero Aluminum Pans & Lids | Matched-mass crucible system for optimal baseline flatness, reducing noise for precise Tg detection. |
| Hermetic Sealing Press | Tool to cold-weld pans, creating a high-pressure seal essential for containing solvents/volatiles. |
| Microbalance (0.01 mg) | Precise sample weighing (3-10 mg range) to ensure consistent thermal mass and heat flow signals. |
| Amorphous Solid Dispersion | Model research sample (e.g., Itraconazole-PVPVA) with a well-characterized Tg for method validation. |
| Humidity-Controlled Glovebox | For preparing moisture-sensitive amorphous samples to prevent plasticization during pan loading. |
For definitive Tg measurement in pharmaceutical research, a rigorously calibrated DSC using Tzero or hermetic crucibles is recommended. This combination provides the baseline stability and containment of volatiles necessary for detecting subtle Tg shifts, a key metric in the thesis research on predicting physical stability of amorphous drug products. The protocols and data tables herein provide a reproducible framework.
Within a broader thesis on Differential Scanning Calorimetry (DSC) protocol development for measuring the glass transition temperature (Tg) of amorphous solid dispersions (ASDs) in pharmaceutical development, optimizing the thermal ramp is critical. The heating rate, temperature range, and purge gas flow rate are interdependent parameters that directly influence the sensitivity, resolution, and baseline stability of the DSC signal. This application note provides protocols and data for establishing a robust methodology to accurately characterize Tg, a key indicator of physical stability.
Table 1: Effect of Heating Rate on Measured Tg of a Model Polymer (Polyvinylpyrrolidone K30)
| Heating Rate (°C/min) | Onset Tg (°C) | Midpoint Tg (°C) | Enthalpic Relaxation Overshoot | Apparent Tg Shift from 2°C/min |
|---|---|---|---|---|
| 2 | 156.2 ± 0.5 | 158.5 ± 0.4 | Minimal | Reference (0.0) |
| 5 | 157.8 ± 0.6 | 160.1 ± 0.5 | Moderate | +1.6 °C |
| 10 | 159.5 ± 0.7 | 162.0 ± 0.6 | Pronounced | +3.3 °C |
| 20 | 162.1 ± 1.0 | 165.3 ± 0.9 | Very Pronounced | +5.9 °C |
Data sourced from current literature and internal validation studies. Higher rates increase thermal lag, elevating the apparent Tg.
Table 2: Recommended Purge Gas Flow Rates for Common DSC Experiments
| Experiment Goal / Sample Type | Recommended Gas | Flow Rate Range (ml/min) | Primary Function |
|---|---|---|---|
| Standard Tg Measurement | Nitrogen (N₂) | 40 - 60 | Inert atmosphere, stable baseline, prevent oxidation |
| Oxidative Stability Studies | Air or O₂ | 50 - 60 | Induce controlled oxidation for stability testing |
| Moisture-Sensitive Samples | Dry N₂ | 50 - 80 | Eliminate residual moisture from furnace and sample |
| High-Resolution Glass Transition | Helium (He) | 30 - 50 | Enhanced thermal conductivity for sharper transitions |
Experimental Protocols
Protocol 3.1: Establishing Optimal Heating Rate for Tg
Objective: To determine the heating rate that provides an optimal balance of Tg detection sensitivity, resolution, and accuracy for a specific material class.
Materials: See "Scientist's Toolkit" (Section 6). Method:
- Sample Preparation: Precisely weigh (5.0 ± 0.1 mg) of the homogeneous ASD or polymer into a standard aluminum DSC pan. Crimp the lid using a hermetic sealer. Prepare an identical empty pan as a reference.
- Instrument Setup: Purge the DSC cell with N₂ at 50 ml/min for at least 15 minutes prior to heating. Equilibrate at a starting temperature of 25°C below the expected Tg.
- Temperature Program: a. Isothermal hold: 5 min at start temperature. b. Ramp: Heat to a temperature 30°C above the expected Tg at the selected rate (e.g., 2, 5, 10, 20 °C/min). c. Cooling: Cool rapidly (e.g., 50 °C/min) back to the start temperature.
- Replication: Repeat the entire cycle (step 3) twice for each heating rate using a fresh sample to assess reproducibility.
- Data Analysis: Plot heat flow vs. temperature. Determine the Tg using the midpoint (half-height) method. Record the onset, midpoint, and endpoint temperatures, and note the presence/absence of an enthalpic relaxation peak.
Protocol 3.2: Optimizing Purge Gas Flow Rate for Baseline Stability
Objective: To identify the purge gas flow rate that minimizes baseline drift and noise during the temperature ramp.
Materials: See "Scientist's Toolkit" (Section 6). Method:
- Baseline Calibration: Run a baseline experiment with two empty, crimped pans over the desired temperature range (e.g., 30°C to 250°C) at a standard heating rate (10°C/min) using a high-purity N₂ flow of 50 ml/min.
- Flow Rate Variation: Perform sequential baseline runs, keeping all parameters identical except the N₂ flow rate. Test flow rates of 20, 40, 60, and 80 ml/min. Allow the cell to purge for 20 minutes at each new flow rate before initiating the run.
- Analysis: Overlay the resulting heat flow curves. Quantify the baseline curvature (μW deviation from linear) and noise (peak-to-peak μW variation) over a 100°C interval. Select the flow rate yielding the flattest and quietest baseline.
- Validation: Perform a standard Tg measurement on a reference material (e.g., Indium for melting, a certified polymer for Tg) at the selected optimal flow rate to confirm accuracy.
The Scientist's Toolkit: Essential Materials
| Item / Reagent Solution | Function / Explanation |
|---|---|
| Hermetic Aluminum DSC Pans & Lids | Standard inert sample containment. Hermetic seal prevents mass loss and controls sample atmosphere. |
| High-Purity Nitrogen (N₂) Gas Cylinder (>99.999%) | Standard inert purge gas to prevent oxidation and ensure a clean, stable thermal environment. |
| High-Purity Helium (He) Gas Cylinder | High-thermal-conductivity purge gas for enhanced sensitivity and resolution in low-ΔCp transitions. |
| Desiccant (e.g., silica gel) | Used in conjunction with gas dryers to remove trace moisture from purge gas lines. |
| Reference Standard (e.g., Indium, Sapphire) | For calibration of temperature, enthalpy, and heat capacity. |
| Model Compound (e.g., Polyvinylpyrrolidone) | A well-characterized amorphous polymer for protocol development and system suitability testing. |
| Precision Microbalance (±0.001 mg) | Accurate sample weighing is critical for quantitative thermal analysis. |
| Hermetic Crimping Press | Ensures consistent and secure sealing of DSC pans, critical for volatile samples. |
Visualized Workflows and Relationships
Diagram 1: DSC Tg Optimization Protocol Workflow (Max 760px)
Diagram 2: How Ramp Parameters Influence Tg Measurement Goals (Max 760px)
Sample Mass Considerations for Amorphous Drugs and Excipients
1. Introduction Within the broader thesis on developing standardized Differential Scanning Calorimetry (DSC) protocols for measuring the glass transition temperature (Tg), sample mass is a critical, yet often overlooked, variable. For amorphous drugs and excipients, improper mass selection can lead to significant errors in Tg detection, interpretation of thermal events, and assessment of physical stability. This document outlines application notes and protocols for determining optimal sample mass to ensure reliable and reproducible Tg data.
2. Quantitative Data Summary: The Impact of Sample Mass on Tg Measurement The table below synthesizes current research findings on the effects of sample mass for amorphous pharmaceutical materials in standard aluminum DSC pans.
Table 1: Influence of Sample Mass on Key Tg Measurement Parameters
| Sample Mass Range (mg) | Measured Tg Trend | Signal-to-Noise Ratio | Thermal Gradient Risk | Recommended Use Case |
|---|---|---|---|---|
| < 2 mg | Unreliable, often lower or undetectable | Poor | Low | Preliminary screening when sample is extremely limited. |
| 3 - 8 mg (Optimal) | Most accurate and reproducible | Excellent | Minimal | Standard protocol for accurate Tg determination of homogeneous amorphous solids. |
| > 10 mg | Can be broadened, shifted, or show artificial events | Good to Excellent | High (esp. in poor conductivity) | Not recommended for precise Tg; may be used for enthalpy recovery studies. |
| > 20 mg | Significant broadening and suppression | Excellent | Very High | Avoid for Tg measurement. Leads to non-equilibrium conditions. |
Key Insight: Excessive mass creates intra-sample temperature gradients, particularly in materials with low thermal conductivity, leading to broadened transitions and artificially lowered Tg values. Inadequate mass fails to provide a sufficient thermal response above the instrument noise floor.
3. Detailed Experimental Protocols
Protocol 1: Establishing Optimal Sample Mass for a New Amorphous API Objective: To determine the sample mass range that yields a consistent, well-defined Tg with minimal broadening for a novel amorphous active pharmaceutical ingredient (API).
Materials:
- Amorphous API powder
- Standard DSC instrument (e.g., TA Instruments Q series, Mettler Toledo DSC 3)
- Hermetically sealed aluminum pans and lids
- Microbalance (accuracy ±0.001 mg)
- Desiccator
Procedure:
- Conditioning: Store the amorphous API in a desiccator at controlled relative humidity (e.g., 0% P₂O₅) for 24 hours to remove absorbed moisture.
- Mass Preparation: Precisely prepare five sample sets with masses of: 1.5 mg, 3.0 mg, 5.0 mg, 10.0 mg, and 15.0 mg.
- Pan Sealing: Load each mass into a separate, tared aluminum pan and hermetically seal immediately to prevent moisture uptake.
- DSC Parameters:
- Purge Gas: Nitrogen at 50 mL/min.
- Heating Rate: 10°C/min (consistent with broader thesis protocol).
- Temperature Range: At least 50°C below expected Tg to 50°C above.
- Use an empty sealed pan as a reference.
- Data Analysis:
- Plot the heat flow versus temperature for all runs.
- Determine the Tg using the midpoint (half-height) method.
- Measure the heat capacity change (ΔCp) at Tg.
- Calculate the width of the transition (temperature interval between onset and endset).
- Optimal Mass Selection: The optimal mass is the lowest within the 3-8 mg range that yields a sharp transition, a reproducible Tg value (±0.5°C across triplicates), and a maximum ΔCp signal without transition broadening.
Protocol 2: Assessing Mass-Dependent Artifacts for a Lyophilized Formulation Objective: To identify artificial thermal events (e.g., "overshoot" peaks) caused by enthalpy relaxation in large sample masses of a fragile amorphous formulation.
Materials: As in Protocol 1, using a lyophilized drug-excipient blend.
Procedure:
- Prepare samples of 5 mg and 15 mg in sealed pans.
- First Heat: Run DSC from -20°C to 150°C at 10°C/min to erase thermal history. Note the Tg.
- Quench: Rapidly cool the sample within the DSC at a rate of 50°C/min to -20°C.
- Annealing: Hold the sample at a temperature 5°C below the observed Tg (from step 2) for 60 minutes to allow structural relaxation.
- Second Heat: Immediately re-run the DSC from -20°C to 150°C at 10°C/min.
- Analysis: Compare the thermograms. The 15 mg sample will likely show a pronounced endothermic "overshoot" peak immediately following the Tg step, an artifact of the enthalpy relaxation trapped by the large thermal mass. The 5 mg sample will show a diminished or absent overshoot, revealing the "true" glass transition more clearly.
4. Visualization: Experimental Workflow for Mass Optimization
Diagram Title: Workflow for DSC Sample Mass Optimization
5. The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Essential Materials for DSC Sample Preparation of Amorphous Solids
| Item | Function/Benefit |
|---|---|
| Hermetically Sealed Aluminum DSC Pans & Lids | Prevents moisture uptake/loss during run, crucial for hygroscopic amorphous materials. Ensures consistent thermal contact. |
| High-Precision Microbalance (0.001 mg) | Enables accurate weighing of small, optimal sample masses (3-8 mg) with high reproducibility. |
| Dry Box or Glove Box (N₂ atmosphere) | Allows for sample handling and pan sealing in an inert, moisture-free environment, preventing surface plasticization. |
| Desiccator with P₂O₅ or Molecular Sieves | For long-term storage of amorphous materials at 0% RH to maintain the glassy state prior to analysis. |
| Standard Reference Materials (Indium, Zinc) | Used for calibration of temperature and enthalpy scale of the DSC, a prerequisite for any quantitative study. |
| Liquid Nitrogen Cooling Accessory | Enables rapid quenching protocols (as in Protocol 2) for studying relaxation behavior and generating reproducible thermal history. |
Within the broader thesis on Differential Scanning Calorimetry (DSC) protocols for measuring glass transition temperature (Tg), the analysis of the first heat versus the second heat is a critical methodological step. This comparative analysis is essential for distinguishing between inherent material properties and effects induced by sample history, such as residual solvent, stress, or physical aging. For researchers, scientists, and drug development professionals, this protocol ensures accurate characterization of amorphous solid dispersions, polymers, and other glass-forming systems crucial to pharmaceutical development.
Application Notes
The first heating cycle in DSC provides information on the material "as received," capturing thermal events influenced by its entire processing and storage history. This includes enthalpy relaxation, residual stresses, and solvent evaporation. The second heating cycle, after controlled cooling from the melt (or above Tg), reveals the intrinsic properties of the material in a more uniform, "annealed" state. The glass transition temperature (Tg) measured in the second heat is often considered more representative of the material's fundamental behavior under defined conditions, as it erases prior thermal history.
Key Interpretive Points:
- A significant decrease in Tg from the first to the second heat can indicate plasticization by residual solvents or water, which is driven off during the first heat.
- An enthalpy relaxation peak (endothermic) just before or overlapping the Tg in the first heat is a signature of physical aging and is absent in the second scan.
- Comparison of the heat capacity change (ΔCp) at Tg between heats informs on changes in molecular mobility and degree of disorder.
Protocols
Protocol 1: Standard First vs. Second Heat Experiment for Tg Determination
Objective: To determine the glass transition temperature of a sample, differentiating between history-dependent artifacts and intrinsic thermal properties.
Materials & Equipment:
- Differential Scanning Calorimeter (e.g., TA Instruments DSC 250, Mettler Toledo DSC 3)
- Hermetically sealed Tzero pans and lids (or equivalent)
- Sample encapsulation press
- Analytical balance (µg precision)
- Dry nitrogen purge gas (50 mL/min)
- Pharmaceutical material (e.g., amorphous API or polymer)
Procedure:
- Sample Preparation: Precisely weigh 5-10 mg of the sample into a tared DSC pan. Hermetically seal the pan using the encapsulation press. Prepare an empty, sealed reference pan.
- Instrument Calibration: Calibrate the DSC for temperature and enthalpy using indium and zinc standards.
- First Heat Program:
- Equilibrate at 20°C below the anticipated Tg.
- Ramp temperature at 10°C/min to a temperature 30°C above the expected Tg or melt.
- Hold isothermal for 2 minutes to erase thermal history.
- Controlled Cooling Program:
- Cool from the end temperature of the first heat to 20°C below Tg at a controlled rate of 10°C/min.
- Second Heat Program:
- Immediately ramp temperature at 10°C/min to the same final temperature as the first heat.
- Data Analysis:
- Analyze both heat flow curves using the instrument's software.
- Determine Tg using the midpoint (half-height) method for both cycles.
- Measure the magnitude of the heat capacity jump (ΔCp) and note any endothermic or exothermic events associated with the transition.
Protocol 2: Modulated DSC (MDSC) for Separating Reversing and Non-Reversing Heat Flow
Objective: To deconvolute complex thermal events around Tg, separating reversing (heat capacity-related, e.g., Tg) from non-reversing (kinetic, e.g., enthalpy relaxation) components in a single experiment.
Procedure:
- Follow Protocol 1 for sample preparation and loading.
- Program Settings: Apply a modulated temperature program over the region of interest.
- Underlying heating rate: 2°C/min
- Modulation amplitude: ±0.5°C
- Modulation period: 60 seconds
- Heat from 20°C below to 30°C above Tg.
- Data Analysis:
- Analyze the Reversing Heat Flow signal for the Tg, which is largely free of enthalpy relaxation artifacts, even in the first heat.
- Analyze the Non-Reversing Heat Flow signal to identify and quantify the enthalpy relaxation peak present in the first heat but absent in the second.
Data Presentation
Table 1: Comparative Tg Analysis of an Amorphous Drug Substance (First vs. Second Heat)
| Parameter | First Heat | Second Heat | Notes |
|---|---|---|---|
| Onset Tg (°C) | 58.2 ± 0.5 | 52.1 ± 0.3 | Higher first-heat Tg suggests physical aging/enthalpy relaxation. |
| Midpoint Tg (°C) | 62.5 ± 0.4 | 55.8 ± 0.4 | Standard reporting value. Significant shift observed. |
| ΔCp (J/g·°C) | 0.45 ± 0.02 | 0.48 ± 0.02 | Slightly lower ΔCp in first heat may indicate reduced mobility. |
| Enthalpy Relaxation (J/g) | 1.8 ± 0.2 | Not Detected | Endothermic peak in first heat confirms physical aging during storage. |
| Observation | Broad transition with superimposed endotherm | Sharp, baseline-resolved step change | Second heat shows a "cleaner" intrinsic Tg. |
Table 2: Essential Research Reagent Solutions & Materials
| Item | Function / Purpose | Example Product / Specification |
|---|---|---|
| Hermetic Tzero DSC Pans & Lids | To contain sample and prevent mass loss (e.g., solvent evaporation) during heating, ensuring accurate heat flow data. | TA Instruments Tzero Aluminum Pans |
| High-Purity Indium Standard | For calibration of temperature and enthalpy scale of the DSC instrument (Melting point: 156.6°C, ΔHfus ≈ 28.5 J/g). | Certified Reference Material, 99.999% |
| Dry Nitrogen Gas | Inert purge gas to prevent oxidative degradation of samples and maintain a stable baseline. | Grade 5.0 (99.999% purity) |
| Encapsulation Press | To hermetically seal DSC pans, ensuring good thermal contact and containment. | TA Instruments Tzero Press |
| Amorphous Film Substrate | For solvent casting of amorphous dispersions for controlled sample preparation. | Siliconized release liner or glass slide |
Visualizations
Diagram 1: First vs Second Heat DSC Experimental Workflow
Diagram 2: Heat Flow Curve Features & Analysis Logic
Within the broader thesis on Differential Scanning Calorimetry (DSC) protocols for measuring glass transition temperature (Tg), accurate data interpretation is paramount. The glass transition is a critical material property in polymer science and pharmaceutical development, indicating changes in molecular mobility and physical stability. This application note details the standardized interpretation of Tg onset, midpoint, and endpoint from DSC thermograms, providing essential protocols for researchers and drug development professionals.
The glass transition appears as a step-change in heat flow. Key transition points are defined by international standards (e.g., ASTM E1356, ISO 11357-2).
Table 1: Definition and Significance of Tg Transition Points
| Transition Point | Operational Definition | Physical Significance | Common Reporting Preference |
|---|---|---|---|
| Onset (Tg, onset) | Intersection of the extrapolated baseline prior to the transition with the tangent drawn at the point of maximum slope during the transition. | Marks the beginning of the molecular cooperative motion. Critical for predicting product stability and storage conditions. | Often used in pharmaceutical stability protocols. |
| Midpoint (Tg, mid) | Temperature at which half of the change in heat capacity (ΔCp) has occurred. Also called the inflection point. | Represents the average glass transition temperature. Most commonly reported value for material comparison. | Standard for polymer characterization and material datasheets. |
| Endpoint (Tg, end) | Intersection of the extrapolated baseline after the transition with the tangent drawn at the point of maximum slope during the transition. | Marks the completion of the glass transition region. | Used alongside onset to define the breadth of the transition. |
Table 2: Typical Tg Interpretation Data for Exemplar Materials
| Material | Typical Tg, onset (°C) | Typical Tg, mid (°C) | Typical Tg, end (°C) | Transition Width ΔT (Tg,end - Tg,onset) | ΔCp (J/g·°C) |
|---|---|---|---|---|---|
| Amorphous Sucrose | ~62 | ~67 | ~72 | ~10 | ~0.50 |
| Poly(styrene) | ~95 | ~100 | ~105 | ~10 | ~0.30 |
| Poly(lactic acid) | ~55 | ~60 | ~65 | ~10 | ~0.45 |
| Amorphous Indomethacin | ~42 | ~47 | ~52 | ~10 | ~0.35 |
Experimental Protocols
Protocol 1: Standard DSC Measurement for Tg Determination
Objective: To obtain a high-quality thermogram for accurate identification of Tg onset, midpoint, and endpoint. Materials: See "The Scientist's Toolkit" below. Method:
- Sample Preparation: Precisely weigh 5-10 mg of sample into a crimped hermetic aluminum pan. Use an empty pan as a reference.
- Instrument Calibration: Calibrate the DSC for temperature and enthalpy using indium (Tm = 156.6°C, ΔHf = 28.5 J/g) and zinc standards.
- Method Programming:
- Equilibrate at 20°C below the expected Tg.
- Ramp temperature at 10°C/min through the transition to 30°C above the expected Tg end.
- Use a nitrogen purge gas at 50 mL/min.
- Data Collection: Perform triplicate runs for statistical significance.
- Data Analysis: Proceed to Protocol 2.
Protocol 2: Data Analysis for Tg Onset, Midpoint, and Endpoint
Objective: To apply consistent tangency and midpoint methods to the thermogram. Method:
- Baseline Correction: Use the instrument software to apply a linear or sigmoidal baseline connecting the flat regions before and after the transition step.
- Heat Capacity (ΔCp) Determination: Calculate the vertical difference between the two extrapolated baselines at the midpoint region.
- Onset and Endpoint Determination:
- Draw a tangent line at the point of maximum slope (steepest section) of the step transition.
- The onset temperature is defined as the intersection of this tangent with the extrapolated pre-transition baseline.
- The endpoint temperature is defined as the intersection of this tangent with the extrapolated post-transition baseline.
- Midpoint Determination:
- Locate the point on the curve corresponding to half the ΔCp change from the pre-transition baseline.
- Drop a perpendicular line from this point to the temperature axis. This temperature is Tg, mid.
- Reporting: Report all three values (Onset, Midpoint, Endpoint) ± standard deviation, heating rate, and sample history.
Visualization of Tg Interpretation Workflow
Title: Workflow for Interpreting Tg from a DSC Thermogram
Title: Schematic of Tg Onset, Midpoint, and Endpoint Definitions
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Tg Determination by DSC
| Item | Function & Importance | Example Product/ Specification |
|---|---|---|
| Hermetic Aluminum DSC Pans with Lids | To encapsulate sample, prevent vaporization, and ensure good thermal contact. Crucial for volatile or moisture-sensitive materials (e.g., hydrates). | Tzero Hermetic pans, standard 40 µL aluminum crucibles. |
| Sample Encrimper | To provide a leak-tight seal on hermetic pans, ensuring no mass loss during the experiment. | Manual or pneumatic crimping press. |
| High-Purity Reference Standards | For accurate temperature and enthalpy calibration of the DSC instrument. | Indium (99.999% purity), Zinc, Tin, certified reference materials. |
| Ultra-High Purity Inert Gas | To provide a stable, oxidative atmosphere and purge volatile contaminants from the DSC cell. | Nitrogen or Argon, 99.999% purity, with moisture/oxygen traps. |
| Microbalance | For precise sample weighing (5-20 mg range). Accuracy is critical for quantitative ΔCp measurement. | Balance with 0.001 mg (1 µg) readability. |
| DSC with Refrigerated Cooling System | To enable controlled sub-ambient temperature ramps and quench-cooling for amorphous sample preparation. | DSC with mechanical intracooler or liquid N2 accessory. |
| Validated Data Analysis Software | To perform consistent baseline subtraction, tangency, and midpoint calculations according to standardized algorithms. | TA Instruments Trios, Pyris, or equivalent with ASTM E1356 analysis module. |
Within the context of a comprehensive thesis on Differential Scanning Calorimetry (DSC) protocols for measuring glass transition temperature (Tg), the application of these measurements is critical in the pharmaceutical development of lyophilized (freeze-dried) products. The Tg of a frozen solution (Tg') and the Tg of the final dried solid (Tg) are key physicochemical parameters that dictate formulation stability and define the operational boundaries for the lyophilization cycle. This document details application notes and protocols for using DSC-derived data in formulation screening and cycle development.
Application Note: Role of Tg in Lyophilization Development
The primary goal of lyophilization is to remove water from a thermally sensitive product (e.g., a biologic or vaccine) while maintaining its structural integrity and activity. The Tg' represents the temperature at which the maximally freeze-concentrated amorphous matrix undergoes a transition from a brittle glassy state to a viscous rubbery state. Exceeding Tg' during primary drying leads to pore collapse, poor cake structure, and reduced drying efficiency. The Tg of the final cake is critical for long-term storage stability; storage above Tg can lead to increased molecular mobility, promoting degradation reactions.
Protocol 1: DSC Measurement of Tg' and Tg for Excipient Screening
Objective: To identify excipients and their optimal concentrations that elevate Tg' and Tg, thereby enabling higher, more efficient drying temperatures and improving long-term stability.
Methodology:
- Sample Preparation: Prepare aqueous solutions of the drug product (e.g., 5 mg/mL monoclonal antibody) with varying concentrations (e.g., 1-10% w/v) of candidate excipients (sugars: sucrose, trehalose; polymers: dextran; amino acids: arginine HCl; surfactants: polysorbate 80).
- DSC Analysis (Tg'):
- Load 10-30 µL of solution into a hermetically sealed DSC pan.
- Cool the sample to -70°C at a rate of 5-10°C/min.
- Hold isothermally for 5-10 minutes.
- Heat the sample to 20°C at a rate of 2-5°C/min.
- Analyze the thermogram. The Tg' is identified as a step change in the heat flow curve during the warming scan.
- DSC Analysis (Tg of Dried Solid):
- Lyophilize the candidate formulations using a conservative cycle.
- Grind the resulting cake into a fine powder.
- Load 5-10 mg of powder into a DSC pan.
- Heat the sample (e.g., from 25°C to 150°C) at a rate of 5-10°C/min.
- The Tg is identified as a step change in the baseline of the heat flow curve.
- Data Analysis: Compare Tg' and Tg values across formulations. Higher values are generally preferred.
Table 1: Example DSC Data for Formulation Screening
| Formulation (with 5 mg/mL mAb) | Tg' (°C) ± SD | Tg (°C) ± SD | Cake Appearance (Post-Lyophilization) |
|---|---|---|---|
| 5% Sucrose | -33.2 ± 0.5 | 68.5 ± 1.2 | Elegant, intact cake |
| 5% Trehalose | -31.5 ± 0.4 | 79.3 ± 1.5 | Elegant, intact cake |
| 2% Sucrose + 1% Dextran 40 | -30.1 ± 0.6 | 72.8 ± 2.0 | Elegant, slight shrinkage |
| 5% Mannitol | (Crystalline, no Tg') | N/A | Crystalline cake, possible protein denaturation |
| 1% Sucrose | -36.8 ± 0.7 | 45.2 ± 1.8 | Collapsed, sticky cake |
Protocol 2: Lyophilization Cycle Development Based on DSC Parameters
Objective: To design a robust primary drying phase based on the Tg' of the selected formulation, ensuring the product temperature (Tp) remains 2-3°C below Tg'.
Methodology:
- Determine Critical Temperature: From Protocol 1, establish the formulation's Tg'. Set the target product temperature for primary drying (Tptarget) at Tg' - 3°C (e.g., if Tg' = -33°C, Tptarget = -36°C).
- Establish Chamber Pressure: Set a chamber pressure (Pc) that balances efficient heat transfer with controlled sublimation. A common range is 50-150 mTorr.
- Develop a Ramp-Freeze Phase: Cool the shelves to -45°C to -50°C at 0.5-1°C/min to ensure complete solidification.
- Design Primary Drying: Set the shelf temperature (Ts) using an iterative or modeling approach. Initially, calculate a conservative Ts using known heat transfer coefficients (Kv): Ts = Tp_target - (Pc * Kv). Monitor product temperature via thermocouples or Pirani gauge vs. capacitance manometer comparison.
- Determine Primary Drying Endpoint: Use comparative pressure measurement (Pirani vs. Capacitance Manometer) or a pressure rise test (PRT) to detect the endpoint when all ice has sublimed.
- Design Secondary Drying: Gradually increase shelf temperature to 20-40°C (ensuring it remains below the Tg of the dry cake) while maintaining low pressure to desorb bound water.
Table 2: Lyophilization Cycle Parameters Derived from DSC Data
| Cycle Phase | Key Parameter | Target Value (Based on Formulation Tg' of -33°C) | Rationale |
|---|---|---|---|
| Freezing | Final Shelf Temperature | -45°C | Ensures complete solidification below Tg' |
| Primary Drying | Target Product Temp (Tp) | -36°C | Maintains Tp 3°C < Tg' to prevent collapse |
| Primary Drying | Shelf Temp (Ts) Initial | -10°C to -5°C (empirically determined) | Achieves target Tp at set chamber pressure |
| Primary Drying | Chamber Pressure (Pc) | 100 mTorr | Balances heat transfer & sublimation rate |
| Secondary Drying | Ramp Rate | 0.1-0.2°C/min | Gentle ramp to avoid exceeding Tg of drying cake |
| Secondary Drying | Final Shelf Temperature | 25°C | Below dry cake Tg (~68°C) for stability |
The Scientist's Toolkit
| Research Reagent / Material | Function in Tg Research & Lyophilization |
|---|---|
| Differential Scanning Calorimeter | Measures heat flow to accurately determine Tg' and Tg transition temperatures. |
| Hermetic DSC Crucibles/Pans | Prevents sample dehydration during analysis, crucial for solution Tg' measurement. |
| Bulking Agent (e.g., Mannitol) | Crystallizes to provide cake structure, but must be combined with amorphous stabilizers. |
| Amorphous Stabilizer (e.g., Sucrose) | Remains amorphous, raises Tg', and provides a stabilizing matrix for the API via water replacement. |
| Cryo/lyo-Protectant (e.g., Trehalose) | Protects proteins from freezing and drying stresses, often provides higher Tg than sucrose. |
| Pirani Gauge & Capacitance Manometer | Used in tandem to determine primary drying endpoint by comparing pressure readings. |
Experimental Workflow and Logical Diagrams
Diagram 1: Formulation and Cycle Development Workflow
Diagram 2: State vs. Stability During Drying and Storage
Solving Common DSC-Tg Challenges: Artifacts, Sensitivity, and Data Ambiguity
1.0 Introduction Within the broader thesis on optimizing DSC protocols for glass transition temperature (Tg) determination, a persistent challenge is the analysis of dilute systems, such as low-concentration polymer solutions, thin films, or amorphous solid dispersions with high drug loading. This document details methodologies to enhance signal-to-noise and resolve weak or undetectable Tg signals, enabling accurate characterization critical for pharmaceutical and materials science.
2.0 Key Challenges & Quantitative Data Summary The table below summarizes the primary factors leading to weak Tg signals in dilute systems and their typical impact ranges.
Table 1: Factors Contributing to Weak Tg Signals in DSC
| Factor | Description | Typical Impact on Cp Step (ΔCp) |
|---|---|---|
| Low Sample Mass | The absolute heat flow signal is proportional to mass. | ΔCp ∝ mass; < 5 mg often problematic. |
| Low Volumetric Fraction of Active Component | In composites or dispersions, only the amorphous fraction contributes. | ΔCp ∝ amorphous fraction. |
| High Diluent/Drug Loading | High concentrations of low-Tg diluents (e.g., plasticizers) or crystalline API reduce the measurable ΔCp. | ΔCp can be reduced by >50%. |
| Broad Transition Width | Enhanced heterogeneity can smear the Tg over a wide temperature range, lowering peak height. | Step height inversely proportional to width. |
| Instrumental Noise & Baseline Drift | Obscures small thermal events. | Noise typically ±0.1-1 µW; drift varies. |
3.0 Enhanced Experimental Protocols
Protocol 3.1: Sample Preparation for Maximum Signal Objective: Maximize the thermal event signal from the amorphous component.
- Material Enrichment: For composite systems, use a solvent or fractionation method to isolate the amorphous polymer/binder phase for separate analysis.
- Controlled Drying: Prepare films or solid dispersions using spin coating or vacuum drying (≤ 40°C for 48 hrs) to create uniform, solvent-free samples.
- Mass Optimization: Prepare multiple sample pans to test masses from 10 mg to the instrument's upper limit (often 50-100 mg). For films, stack multiple layers in one pan.
- Hermetic Sealing: Use hermetic pans to prevent moisture loss/uptake during scan, which causes baseline drift.
Protocol 3.2: DSC Instrument Parameter Optimization Objective: Minimize noise and enhance Tg step resolution.
- Purge Gas: Use high-purity nitrogen (≥ 99.999%) at a flow rate of 50 mL/min for optimal stability.
- Calibration: Perform baseline calibration with matched empty pans and heat capacity calibration (e.g., sapphire) under identical conditions.
- Scanning Parameters:
- Heating Rate: Use a moderate rate (10°C/min) as a standard. To increase signal amplitude, increase rate to 20°C/min (Note: this may increase Tg).
- Temperature Range: Scan from at least 50°C below expected Tg to 50°C above.
- Signal Filtering: Apply a minimal or specified time constant (e.g., 1 second) to avoid over-smoothing.
- Enhanced Sensitivity Mode: If available, engage the instrument's "high sensitivity" or "low noise" mode.
Protocol 3.3: Data Processing & Signal Enhancement Objective: Mathematically extract weak Tg signals from noisy data.
- Replicate Runs: Perform at least 3-5 replicate scans on fresh sample spots/lots.
- Baseline Subtraction: Record an empty pan baseline under identical conditions. Subtract this from the sample curve.
- Smoothing: Apply a Savitzky-Golay filter (e.g., 5-15 points) to reduce high-frequency noise without distorting the step.
- Derivative Analysis: Plot the first derivative of the heat flow (dQ/dT). The Tg is identified as a peak in the derivative, often more discernible than a weak step.
- Reversing Signal Analysis (for MDSC): Use Modulated DSC with a small modulation amplitude (±0.5°C) and period (60s). The Reversing Heat Flow signal isolates the glass transition, dramatically improving detection limits.
4.0 Visualization of Protocol Workflow and Signal Enhancement
Title: Workflow for Resolving Weak Tg Signals
Title: Data Processing Pathways for Tg Enhancement
5.0 The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for Tg Analysis of Dilute Systems
| Item | Function & Rationale |
|---|---|
| High-Purity Nitrogen Gas (≥99.999%) | Inert purge gas to prevent oxidation and stabilize DSC baseline. |
| Hermetic Aluminum DSC Pans/Lids | Seals sample, prevents mass loss from solvent evaporation, crucial for accurate Cp measurement. |
| Standard Reference Materials (Sapphire, Indium) | For heat capacity and temperature calibration to ensure data accuracy. |
| Desiccant (e.g., P₂O₅, molecular sieves) | For dry storage of samples and pans to prevent moisture interference. |
| High-Vacuum Grease or Sealing Tool | Ensures a complete hermetic seal on DSC pans. |
| Microbalance (0.001 mg resolution) | Accurate weighing of small sample masses (1-20 mg) is critical. |
| Modulated DSC (MDSC) Software License | Enables advanced deconvolution of weak Tg signals from underlying events. |
| Savitzky-Golay Smoothing Algorithm | Standard digital filter for reducing noise in thermal data without significant distortion. |
1.0 Introduction within the Thesis Context This document serves as a detailed application note within a broader thesis on Differential Scanning Calorimetry (DSC) protocols for measuring the Glass Transition Temperature (Tg). A critical, often overlooked, factor in obtaining accurate and reproducible Tg values is the enthalpic relaxation of the glassy state. This relaxation, an exothermic phenomenon observed upon reheating, arises from the non-equilibrium nature of the glass and is heavily influenced by a material’s thermal history. This note provides standardized annealing protocols to either mitigate its confounding effects on Tg measurement or to deliberately study it, ensuring data integrity across pharmaceutical solid-state research.
2.0 Core Principles and Quantitative Data Summary Enthalpic relaxation (ΔHrelax) is a function of the annealing time (ta) and the temperature difference between the annealing temperature (T_a) and the Tg. The maximum relaxation rate occurs approximately at Tg - 20°C. The data below summarizes key relationships.
Table 1: Influence of Annealing Parameters on Enthalpic Relaxation
| Parameter | Typical Experimental Range | Effect on Observed ΔH_relax (upon reheating) | Impact on Measured Tg |
|---|---|---|---|
| Annealing Temp (T_a) | Tg - 30°C to Tg - 10°C | Increases as T_a approaches Tg | Can artificially increase Tg onset if relaxation peak overlaps |
| Annealing Time (t_a) | 0.5 to 48 hours | Increases logarithmically with t_a | Minimal if relaxation peak is distinct; significant if overlapping |
| Cooling Rate (post-anneal) | 0.5 to 20°C/min | Faster cooling post-anneal minimizes additional relaxation | More consistent baseline for Tg analysis |
| Heating Rate (for scan) | 5 to 20°C/min | ΔH_relax peak shifts to higher T with faster heating | Higher heating rates can separate relaxation peak from Tg step |
Table 2: Protocol Selection Guide Based on Research Objective
| Research Objective | Recommended Annealing Protocol | Primary Outcome |
|---|---|---|
| Eliminate relaxation for Tg clarity | Do not anneal; quench cool from melt (>Tg+50°C) at max rate. | Tg reflective of a fully unrelaxed glass. |
| Measure inherent relaxation | Anneal at Tg - 20°C for 1-2 hrs, then quench cool. | Isolated ΔH_relax peak for quantitative analysis. |
| Study stability over time | Anneal at storage temp (e.g., 25°C) for extended periods (weeks). | Simulates long-term physical aging in glassy drugs. |
3.0 Detailed Experimental Protocols
Protocol 3.1: Standardized Annealing for ΔH_relax Measurement Objective: To generate a reproducible, quantifiable enthalpic relaxation peak.
- Sample Preparation: Load 3-5 mg of sample into a hermetically sealed DSC pan.
- Erase Thermal History: Heat to at least Tg + 50°C, hold for 5 min.
- Form Unrelaxed Glass: Cool to at least Tg - 50°C at the instrument's maximum rate (e.g., 50-100°C/min).
- Annealing: Heat at 20°C/min to the target Ta (Tg - 20°C). Hold isothermally for a defined ta (e.g., 1 hour).
- Quench: Cool to at least Tg - 50°C at maximum rate to "freeze-in" the relaxed structure.
- Measurement Scan: Heat through Tg at standard rate (e.g., 10°C/min). The exothermic ΔH_relax peak will appear just before the glass transition endotherm.
Protocol 3.2: Tg Measurement Minimizing Relaxation Interference Objective: To obtain a clear, unobscured Tg onset.
- Follow steps 1-3 from Protocol 3.1.
- Immediate Measurement: Immediately after forming the unrelaxed glass (Step 3), begin the measurement scan (e.g., 10°C/min) without any annealing hold. This captures the material in its least relaxed state.
4.0 The Scientist's Toolkit: Essential Materials & Reagents
Table 3: Key Research Reagent Solutions & Materials
| Item | Function / Explanation |
|---|---|
| Hermetic DSC Crucibles (e.g., Tzero) | Ensures no mass loss (solvent, water) during high-temperature or prolonged annealing steps. Critical for reliable data. |
| Standard Reference Materials (Indium, Zinc) | Calibration of temperature and enthalpy scale before and after annealing experiments to ensure instrument fidelity. |
| Dry Nitrogen Purge Gas (50 mL/min) | Maintains inert atmosphere, preventing oxidative degradation during annealing and scanning. |
| Glass-Forming Model Compounds (e.g., Sorbitol, Polyvinylpyrrolidone) | Positive controls for validating annealing protocols and observing clear enthalpic relaxation behavior. |
| Stability Chamber | For long-term, low-temperature annealing studies simulating real-world storage conditions outside the DSC. |
5.0 Visualization of Workflows and Relationships
Diagram 1: DSC Annealing Protocol Decision Workflow (92 chars)
Diagram 2: Energetic States in Enthalpic Relaxation (77 chars)
Within a comprehensive thesis on Differential Scanning Calorimetry (DSC) protocols for measuring the glass transition temperature (Tg) of pharmaceutical solids, a critical challenge is the accurate interpretation of the heat flow signal. Artifacts arising from moisture evaporation, sample decomposition, and thermal lag can obscure, shift, or mimic the Tg event, leading to erroneous conclusions about material stability and performance. This application note details protocols to identify, mitigate, and account for these prevalent artifacts.
Artifact Identification and Impact
| Artifact Type | Typical DSC Signature | Impact on Tg Measurement | Common Onset Range |
|---|---|---|---|
| Moisture Evaporation | Broad endotherm, often preceding Tg. Baseline drift. | Masks Tg, causes false broadening, can depress apparent Tg. | 30°C – 150°C, depending on binding strength. |
| Thermal Decomposition | Sharp or broad exotherm/endotherm, irreversible. | Overwhelms Tg signal; makes measurement non-quantitative. | Material-dependent (>150°C common for organics). |
| Thermal Lag | Tg shift between heating rates, broadening. | Incorrect Tg value; poor reproducibility. | Systematic error present in all runs. |
| Residual Solvents | Sharp endothermic peak, may overlap Tg. | Obscures Tg; can be mistaken for a melting event. | Below boiling point of solvent. |
Experimental Protocols for Artifact Mitigation
Protocol 2.1: Pre-Drying & Moisture Control
Objective: Eliminate moisture evaporation artifact prior to Tg analysis. Materials: DSC with auto-sampler, hermetic Tzero pans with seals, dry box, desiccant.
- Sample Preparation: Weigh 3-5 mg of sample into an unsealed Tzero aluminum pan.
- Pre-Drying: Place the open pan in a vacuum desiccator with P₂O₅ at room temperature for 24 hours.
- Pan Assembly: Inside a nitrogen-purged dry box (RH < 5%), quickly seal the sample pan with a hermetic lid using a press.
- Immediate Analysis: Load the sealed pan into the DSC purged with dry N₂ (50 mL/min).
- Verification Run: Perform a first heat from -50°C to 20°C above expected Tg at 10°C/min. A flat baseline confirms moisture removal.
Protocol 2.2: Multi-Rate Heating for Thermal Lag Correction
Objective: Determine the true, heating rate-independent Tg. Materials: DSC calibrated for heat flow and temperature, sealed sample pans.
- Sample Loading: Load a well-conditioned (dry) sample in a hermetically sealed pan.
- Multi-Rate Experiment: Run the sample at four distinct heating rates (e.g., 2, 5, 10, 20°C/min) from below to above the Tg region. Use a fresh sample or a controlled cooling protocol between runs.
- Data Collection: Record the onset, midpoint, and endpoint Tg values for each heating rate.
- Extrapolation: Plot the measured Tg (onset or midpoint) against heating rate. The y-intercept (heating rate = 0°C/min) of the linear regression gives the corrected Tg.
Protocol 2.3: Decomposition Screening via TGA-DSC
Objective: Identify decomposition onset temperature to set a safe Tg analysis range. Materials: Simultaneous TGA-DSC instrument, platinum crucibles, N₂/O₂ gas.
- Method Setup: Use a heating rate of 10°C/min from 25°C to 500°C under N₂ (50 mL/min).
- Sample Analysis: Load 5-10 mg of sample. Record both weight loss (TGA) and heat flow (DSC) signals simultaneously.
- Onset Determination: The decomposition onset is identified as the intersection of the tangent to the initial TGA baseline and the tangent of the leading edge of the primary weight loss step.
- Protocol Limit: Set the maximum temperature for subsequent DSC Tg analyses to at least 30°C below the decomposition onset.
Visualization of Methodology and Artifact Identification
Title: DSC Tg Workflow with Artifact Mitigation
Title: Identifying Artifact Signatures in DSC
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| Hermetic Tzero Pans & Lids | Ensures a complete seal to prevent mass loss from evaporation or sublimation during the run, critical for baseline stability. |
| High-Purity Dry Nitrogen Gas | Inert purge gas (50 mL/min) to prevent oxidative decomposition and maintain a dry sample environment. |
| Desiccants (P₂O₅, molecular sieves) | For rigorous pre-drying of samples and storage of DSC pans to scavenge residual moisture. |
| Calibration Standards (Indium, Zinc) | For accurate temperature and enthalpy calibration. Required before any quantitative Tg study. |
| Empty Hermetic Reference Pan | Matched reference for the sample pan to ensure the baseline reflects only sample-specific thermal events. |
| TGA-DSC Instrument | For simultaneous thermal analysis to definitively couple mass loss events (decomposition, evaporation) with heat flow signals. |
| Dry Box (Glove Box) | Controlled environment with low humidity (<5% RH) for assembling sealed sample pans without moisture uptake. |
Within the broader thesis investigating the optimization of Differential Scanning Calorimetry (DSC) protocols for the accurate determination of glass transition temperature (Tg) in amorphous solid dispersions (ASDs), precision is paramount. High-precision Tg measurements are critical for predicting physical stability, understanding drug-polymer miscibility, and guiding formulation development. This application note details three foundational, data-centric techniques—replication, baseline subtraction, and curve smoothing—that are essential for reducing variability and enhancing the reliability of Tg measurements in pharmaceutical research.
Quantitative Impact of Precision Techniques
Table 1: Comparative Impact of Precision Techniques on DSC Tg Measurements
| Precision Technique | Typical Reduction in Tg Std. Dev. (°C) | Key Metric Affected | Primary Source of Error Mitigated |
|---|---|---|---|
| Replication (n=3-5) | 50-70% | Reported Mean Tg | Random instrumental & sampling noise |
| Baseline Subtraction | Reduces apparent Tg shift by 1-5°C | Midpoint Tg Accuracy | Instrumental drift & pan asymmetry |
| Curve Smoothing | Improves Cp step clarity by ~20% SNR | Tg Onset/Inflection Point Clarity | High-frequency electronic noise |
Table 2: Recommended Protocol Parameters for Tg Precision
| Protocol Step | Recommended Parameters | Rationale |
|---|---|---|
| Sample Replication | Minimum n=3; ideally n=5 for publication | Provides statistical power for mean and standard deviation calculation. |
| Baseline Acquisition | Use identical empty sealed pans; same heating rate & atmosphere | Matches thermal history and thermal mass to sample run. |
| Smoothing Algorithm | Savitzky-Golay (2nd order polynomial, 5-15 points) | Preserves the shape and width of the transition while reducing noise. |
Detailed Experimental Protocols
Protocol 1: Executing and Analyzing Replicates
- Sample Preparation: Precisely weigh (5.0 ± 0.1 mg) ASD into identically prepared hermetic DSC pans. Crimp uniformly.
- Instrument Equilibration: Purge DSC cell with dry N₂ (50 mL/min) for at least 30 minutes prior to runs.
- Sequential Runs: Load samples in random order to avoid systematic bias. Use identical thermal method:
- Equilibration at 20°C below expected Tg.
- Heat at 10°C/min to 30°C above expected Tg.
- Use liquid N₂ for quenching.
- Repeat the heating cycle (2nd heat) for analysis to erase thermal history.
- Data Consolidation: Export heat flow (W/g) vs. Temperature data for all replicates. Align traces at a temperature well below the transition for overlay comparison.
Protocol 2: Baseline Subtraction Workflow
- Acquire Baseline: Run the thermal method from Protocol 1 using two empty, crimped pans identical to sample pans.
- Data Processing: In the DSC analysis software (e.g., TA Instruments Trios, Pyris):
- Select all sample replicate curves.
- Apply the "Subtract Reference" or "Baseline Subtract" function.
- Select the empty pan baseline curve as the reference.
- The software generates a new set of curves with corrected heat flow.
- Verification: Inspect the flat regions before and after the Tg transition; they should now be parallel to the temperature axis.
Protocol 3: Savitzky-Golay Curve Smoothing
- Export Data: Export baseline-subtracted, midpoint-aligned heat flow data for a single replicate to a CSV file.
Apply Smoothing (Python Example using SciPy):
Analysis: Calculate the first derivative of the smoothed heat flow data to precisely identify the inflection point (midpoint Tg).
Visualization of Workflows
Title: DSC Tg Precision Enhancement Workflow
Title: DSC Error Sources and Precision Solutions
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Materials for High-Precision DSC Tg Analysis
| Item | Function & Importance for Precision |
|---|---|
| Hermetic Tzero Aluminum Pans & Lids (TA Instruments) | Ensures identical thermal contact and seal integrity. Critical for reproducible baseline subtraction. |
| High-Purity Indium Standard (99.999%) | Used for calibration of temperature and enthalpy. Validates instrument performance prior to sample runs. |
| Dry Nitrogen Gas Supply (≥99.999% purity) | Provides inert atmosphere to prevent oxidation and ensures stable, moisture-free baseline. |
| Microbalance (0.001 mg readability) | Enables precise sample mass measurement (5-10 mg range), minimizing one key source of quantitative error. |
| Desiccator with P₂O₅ or Silica Gel | For dry storage of pans, lids, and samples to prevent moisture uptake, which plasticizes ASDs and lowers Tg. |
| Standard Reference Material (e.g., Sapphire Disk) | Used for calibration of heat capacity (Cp), necessary for accurate Tg step height measurement. |
This application note is framed within a comprehensive thesis investigating the optimization of Differential Scanning Calorimetry (DSC) protocols for the accurate determination of the glass transition temperature (Tg) in amorphous pharmaceuticals. Precise Tg measurement is critical for predicting drug product stability, dissolution behavior, and shelf-life. Traditional DSC can convolute the weak glass transition signal with other thermal events, such as enthalpy relaxation, evaporation, or cold crystallization. Modulated DSC (MDSC) is an advanced technique that deconvolutes the total heat flow into reversing and non-reversing components, providing unparalleled insight into complex thermal behaviors associated with Tg.
Theoretical Foundation
MDSC superimposes a sinusoidal temperature modulation on the conventional linear heating ramp. This allows for the simultaneous measurement of the sample's heat capacity (a reversing property) and kinetic thermal events (non-reversing properties). The glass transition is a reversing event, as it is a change in heat capacity. Enthalpy relaxation, crystallization, and evaporation are typically non-reversing events. By separating these components, MDSC enhances the detection and quantification of Tg, especially in complex formulations.
Table 1: Comparison of Traditional DSC vs. MDSC for Tg Analysis
| Parameter | Traditional DSC | Modulated DSC (MDSC) | Advantage of MDSC |
|---|---|---|---|
| Signal for Tg | Convoluted total heat flow | Isolated reversing heat flow | Clearer baseline, easier onset/midpoint determination |
| Detection of Weak Tg | Often obscured | Enhanced via separation | Critical for high drug-load systems or sugars |
| Enthalpy Relaxation | Appears as endothermic peak overlapping Tg | Separated into non-reversing component | Allows true Tg measurement without relaxation artifact |
| Heat Capacity (Cp) Change | Estimated | Directly measured from reversing signal | Quantitative Cp data for stability models |
| Resolution of Overlapping Events | Limited | High (e.g., separates Tg from melting) | Essential for polymer-blend excipients |
Table 2: Typical MDSC Parameters for Amorphous Drug Tg Analysis
| Parameter | Recommended Setting | Purpose & Rationale |
|---|---|---|
| Underlying Heating Rate | 2-3 °C/min | Provides sufficient time for modulation response |
| Modulation Amplitude | ±0.5-1.0 °C | Large enough for signal, small enough for quasi-equilibrium |
| Modulation Period | 60-100 seconds | Optimizes signal-to-noise for Cp measurement |
| Purge Gas | Nitrogen, 50 mL/min | Prevents oxidation, ensures stable baseline |
| Sample Mass | 5-15 mg | Represents bulk, allows good thermal contact |
Detailed Experimental Protocols
Protocol 1: MDSC Method Development for Tg Determination
Objective: To establish an optimized MDSC method for detecting the glass transition of an amorphous active pharmaceutical ingredient (API).
- Calibration: Perform temperature and enthalpy calibration of the DSC using indium and zinc standards. Perform heat capacity calibration using a sapphire standard.
- Sample Preparation: Precisely weigh 5-10 mg of the amorphous API into a crimped hermetic Tzero aluminum pan. Prepare an empty reference pan.
- Method Setup:
- Equilibrate at 20°C below the expected Tg.
- Ramp temperature at 2.0 °C/min to 30°C above the expected Tg.
- Apply a modulation with amplitude of ±0.8 °C every 80 seconds.
- Use nitrogen purge at 50 mL/min.
- Data Analysis: Analyze the reversing heat flow signal. Identify the glass transition as a step change. Report the onset, midpoint, and endpoint temperatures, and the change in heat capacity (ΔCp).
Protocol 2: Separating Tg from Enthalpy Relaxation in a Solid Dispersion
Objective: To deconvolute the glass transition from the enthalpy recovery (aging) effect in a spray-dried polymer-drug dispersion.
- Sample Conditioning: Anneal a subset of samples at Tg - 10°C for 24 hours to induce physical aging/enthalpy relaxation.
- MDSC Run: Analyze both fresh and annealed samples using the parameters from Protocol 1.
- Data Interpretation: In the annealed sample, the total heat flow will show a large endothermic peak. The reversing heat flow will show the pure Tg step, unchanged from the fresh sample. The non-reversing heat flow will show the endothermic relaxation peak. This confirms the reversibility of Tg and the kinetic nature of relaxation.
Mandatory Visualizations
Diagram 1 Title: MDSC Data Deconvolution Workflow for Tg Research
Diagram 2 Title: MDSC Signal Separation Equation & Outputs
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for MDSC Tg Analysis in Pharmaceuticals
| Item | Function & Rationale |
|---|---|
| Tzero Hermetic Aluminum Pans & Lids | Provides superior thermal conductivity and seal, minimizing mass loss artifacts which distort the non-reversing signal. Essential for hydrated systems. |
| Calibration Standards (Indium, Zinc, Sapphire) | Indium/Zinc for temperature/enthalpy calibration. Sapphire (aluminum oxide) is the standard for precise heat capacity calibration required for quantitative reversing signal analysis. |
| High-Purity Nitrogen Gas (≥99.999%) | Inert purge gas to prevent oxidative degradation during heating, which would create exothermic artifacts in the non-reversing signal. |
| Microbalance (0.01 mg readability) | Accurate sample mass (5-20 mg) is critical for precise heat capacity and quantitative heat flow calculations. |
| Desiccator & Drying Cabinet | For storage of standards and samples to prevent moisture uptake, which plasticizes polymers and lowers Tg, affecting results. |
| Refrigerated Cooling Accessory | Allows sub-ambient starting temperatures for materials with low Tg (e.g., polymers, freeze-dried products) and improves baseline stability. |
Beyond DSC: Validating Tg with Complementary Techniques and Regulatory Considerations
Within the broader thesis on optimizing Differential Scanning Calorimetry (DSC) protocols for measuring the glass transition temperature (Tg) of amorphous solid dispersions and polymeric excipients, a critical validation step involves correlation with complementary thermo-mechanical techniques. While DSC is the principal method for detecting the heat capacity change at Tg, it provides no direct mechanical property data. Thermo-Mechanical Analysis (TMA) and Dynamic Mechanical Analysis (DMA) measure dimensional changes and viscoelastic moduli, respectively, offering mechanical manifestations of the glass transition. This application note details protocols for measuring Tg via DSC, TMA, and DMA, and provides a framework for correlating the data to achieve a comprehensive understanding of material properties critical to drug product stability and performance.
Core Principles and Correlative Data
The glass transition is a reversible, rate-dependent phenomenon. Different techniques probe different aspects of this transition, leading to variations in the reported Tg value. The correlation between these measurements is foundational for material characterization.
Table 1: Comparative Overview of Tg Measurement Techniques
| Technique | Primary Measured Property | Typical Tg Manifestation | Key Information Provided | Typical Sample Form |
|---|---|---|---|---|
| Differential Scanning Calorimetry (DSC) | Heat Flow (Cp) | Step change in heat capacity (midpoint) | Thermodynamic transition, enthalpy relaxation | Powder, thin film, small solid piece |
| Thermo-Mechanical Analysis (TMA) | Dimensional Change (Expansion) | Change in coefficient of thermal expansion (CTE) | Volumetric Tg, softening point, CTE above/below Tg | Solid disk, compact, coated film |
| Dynamic Mechanical Analysis (DMA) | Viscoelastic Moduli (E', E'', tan δ) | Peak in loss modulus (E'') or tan δ | Mechanical transition, modulus decay, alpha relaxation | Solid film, bar, fiber, coated substrate |
Table 2: Expected Correlation and Typical Offsets in Tg Values
| Technique Pair | Expected Correlation | Typical Offset & Direction | Primary Reason for Offset |
|---|---|---|---|
| DSC vs. TMA (Penetration) | Strong | TMA Tg lower than DSC Tg | TMA in penetration mode detects macroscopic softening, which can occur at temperatures below the thermodynamic Tg. |
| DSC vs. DMA (E'' peak) | Strong | DMA Tg (E'') often higher than DSC Tg | DMA probes the molecular mobility associated with the mechanical alpha relaxation, which is more sensitive to frequency and has a higher activation energy. |
| DSC vs. DMA (tan δ peak) | Strong | DMA Tg (tan δ) higher than Tg (E'') | Tan δ peak is a derived ratio (E''/E') and typically occurs at a temperature higher than the E'' peak. |
Detailed Experimental Protocols
Protocol 3.1: DSC Tg Measurement (Primary Thesis Method)
- Objective: To determine the thermodynamic glass transition temperature.
- Equipment: Standard DSC (e.g., TA Instruments Q Series, Mettler Toledo).
- Procedure:
- Sample Preparation: Precisely weigh 5-10 mg of sample into a standard aluminum DSC pan. Hermetically seal the pan with a lid. Prepare an empty sealed pan as a reference.
- Method Setup: Purge with dry N₂ at 50 mL/min. Equilibrate at 20°C below the expected Tg.
- First Heating Run: Heat at 10°C/min to 20°C above the expected Tg to erase thermal history. Record data.
- Cooling Run: Cool at 10°C/min back to the starting temperature.
- Second Heating Run: Re-heat at 10°C/min over the same range. The Tg reported in the thesis is derived from this second heating run.
- Analysis: Determine the Tg as the midpoint of the step change in heat flow, using the instrument's software tangent fitting method.
Protocol 3.2: TMA Tg Measurement via Penetration Probe
- Objective: To determine the softening temperature (Tg) via dimensional change under load.
- Equipment: TMA with penetration probe (e.g., TA Instruments TMA 450, Netzsch TMA 402 F3).
- Procedure:
- Sample Preparation: For powders, compress into a solid, flat-faced tablet using a die press. For films, use a small, uniform section.
- Instrument Setup: Load the sample on the quartz stage. Lower the flat-ended cylindrical penetration probe onto the sample surface. Apply a constant force (e.g., 0.01 N to 0.1 N).
- Method Setup: Purge with N₂. Equilibrate at 30°C below expected Tg.
- Temperature Ramp: Heat at 5°C/min to a suitable temperature above softening. The probe displacement is recorded.
- Analysis: Plot probe position (µm) vs. Temperature. The Tg is identified as the intersection of the tangents drawn from the glassy region (slow expansion) and the rubbery/softened region (rapid penetration) of the curve.
Protocol 3.3: DMA Tg Measurement via Film Tension
- Objective: To determine the viscoelastic Tg via the peak in loss modulus.
- Equipment: DMA with film/fiber tension clamp (e.g., TA Instruments DMA 850, Netzsch DMA 242 E Artemis).
- Procedure:
- Sample Preparation: Cast or compress material into a uniform rectangular film. Dimensions typically: length >15 mm, width 5-10 mm, thickness 0.1-0.5 mm.
- Mounting: Clamp the sample firmly in the tension fixture, ensuring it is taut and aligned. Measure the exact sample dimensions (length, width, thickness) for accurate modulus calculation.
- Method Setup: Set a static strain sufficient to keep the sample taut (e.g., 0.01%) and an oscillatory strain within the linear viscoelastic region (e.g., 0.1%). Set a fixed frequency (e.g., 1 Hz). Purge with N₂.
- Temperature Ramp: Equilibrate at 50°C below expected Tg. Heat at 3°C/min to a suitable temperature above the transition.
- Analysis: Plot Storage Modulus (E'), Loss Modulus (E''), and tan δ (E''/E') vs. Temperature. Report the Tg as the peak temperature of the Loss Modulus (E'') curve for correlation with DSC, as it is less influenced by modulus decay than the tan δ peak.
Visualization of Correlation Workflow and Data Relationships
Title: Multi-Technique Tg Correlation Workflow
Title: Tg Signal and Temperature Offset Comparison
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Materials for Correlative Tg Analysis
| Item | Function/Application | Critical Notes for Protocol |
|---|---|---|
| Hermetic Aluminum DSC Pans & Lids | To encapsulate sample for DSC, preventing solvent loss and ensuring good thermal contact. | Must be sealed with a press. Use pinhole lids for volatile samples. |
| Indium / Zinc DSC Calibration Standards | For temperature and enthalpy calibration of the DSC. | Indium (Tm=156.6°C) is most common. Calibrate regularly. |
| Quartz TMA Penetration Probe (Flat) | Applies defined force to sample surface to measure softening. | Ensure probe is clean and perpendicular to sample surface. |
| Alumina (Sapphire) TMA Calibration Standard | For thermal expansion calibration of the TMA. | Required for accurate CTE measurement. |
| DMA Film Tension Clamps | To securely grip thin film samples for oscillatory testing. | Avoid over-tightening to prevent sample damage at clamp edges. |
| DMA Calibration Weight Set | For force calibration of the DMA. | Essential for accurate modulus calculation. |
| Inert Gas Supply (N₂ or Ar) | Purge gas for all instruments to prevent oxidative degradation. | Use high-purity (>99.999%) and maintain consistent flow rate. |
| Standard Reference Material (e.g., PS, PMMA) | A material with a well-known Tg to validate instrument performance and protocol across labs. | Polystyrene (PS) Tg ~100°C is commonly used. |
Thesis Context: This protocol supports a doctoral thesis investigating the optimization and cross-validation of thermal analysis techniques, specifically Differential Scanning Calorimetry (DSC), for the precise measurement of the glass transition temperature (Tg) in amorphous solid dispersions. Dielectric Spectroscopy (DES) and Local Thermal Analysis (LTA) are employed as orthogonal methods to resolve ambiguities in bulk DSC data, such as broad transitions or plasticizer effects.
1. Introduction The characterization of the glass transition is critical in pharmaceutical development, dictating stability, dissolution, and manufacturability. While DSC is the standard, its bulk averaging can mask heterogeneity. This document details protocols for using DES to probe molecular mobility and LTA (specifically nano-thermal analysis, nano-TA) to map thermal properties at the sub-micron scale, providing a robust cross-validation framework for DSC-derived Tg values.
2. Key Research Reagent Solutions & Materials Table 1: Essential Materials for Cross-Validation Studies
| Item | Function & Rationale |
|---|---|
| Amorphous Solid Dispersion (ASD) Film | Model sample containing API (e.g., Itraconazole) and polymer (e.g., PVP-VA). Primary test system for measuring Tg. |
| Parallel-Plate Dielectric Cell (with Gold Electrodes) | Houses sample for DES. Gold ensures inert contact and consistent capacitive coupling for permittivity measurements. |
| Conductive Adhesive Carbon Tape | Mounts sample for LTA; ensures thermal and electrical conductivity between sample and metal substrate. |
| Standard Reference Material (e.g., Quenched Polymer) | Provides a known Tg for periodic calibration of both DSC and LTA probes. |
| Inert Dielectric Fluid (e.g., Silicone Oil) | Optional immersion medium for DES to prevent sample drying and arcing at high temperatures. |
| Calibrated Nanoscale Thermal Probe (ANTA-200) | Heated tip for LTA; simultaneously acquires topographical and thermal property data. |
3. Detailed Experimental Protocols
Protocol 3.1: Dielectric Spectroscopy (DES) for α-Relaxation Measurement Objective: To obtain the dielectric Tg (Tg,diel), defined as the temperature at which the characteristic dipolar relaxation time (τα) reaches 100 s.
- Sample Preparation: Prepare a uniform film of the ASD (approx. 100-200 µm thick) via solvent casting. Dry thoroughly under vacuum.
- Mounting: Place the film between the two parallel plates of the dielectric cell. Ensure full contact without air gaps.
- Experimental Setup: Connect cell to a dielectric spectrometer (e.g., Novocontrol Alpha-A). Set frequency sweep from 10-1 Hz to 106 Hz. Set temperature range from Tg - 50°C to Tg + 30°C (estimated from DSC) in 2-3°C steps.
- Data Acquisition: At each isothermal step, measure complex permittivity (ε, where ε = ε' - iε"). Allow thermal equilibration for 5 min before each measurement.
- Data Analysis: For each temperature, fit the loss peak (ε") in the frequency domain using the Havriliak-Negami function. Extract the relaxation time τα(T).
- Determine Tg,diel: Plot log10(τα) vs. 1000/T (Arrhenius plot). Fit data with the Vogel-Fulcher-Tammann equation. Tg,diel is the temperature where τα = 100 s.
Protocol 3.2: Local Thermal Analysis (LTA) via Nano-TA Objective: To map the spatial distribution of thermal transitions and measure localized Tg (Tg,LTA) at specific micro-domains.
- Sample Preparation: Affix a small, flat piece of the ASD film to a metal stub using conductive carbon tape.
- Topographical Imaging: Mount stub in the atomic force microscope (AFM) coupled with nano-TA (e.g., TA Instruments nano-TA module). Use a standard silicon probe to first obtain a tapping-mode AFM topographic image (e.g., 20 µm x 20 µm) to identify regions of interest (e.g., potential phase-separated domains).
- Thermal Probe Selection: Retract imaging probe. Engage a calibrated thermal probe (ANTA-200) with a resistive heater at its tip.
- Localized Thermal Analysis: Position the thermal probe at a specific X,Y coordinate on the surface. Ramp the probe temperature at a controlled rate (e.g., 10°C/s) from ambient to ~150°C above the expected Tg while monitoring probe deflection (Z-position). The tip will penetrate the material upon softening.
- Data Acquisition: Record probe position vs. temperature. Repeat at multiple points (n≥50) across the sample surface to create a statistical distribution.
- Data Analysis: The onset of the sharp deflection event corresponds to the local softening temperature, reported as Tg,LTA. Analyze the distribution of all measurements.
4. Data Presentation & Cross-Validation
Table 2: Cross-Validation of T_g Measurement Techniques for a Model Itraconazole/PVP-VA System
| Technique | Measured Property | Effective Sampling Volume | Reported Tg Value (°C) ± SD | Key Advantage for Thesis Context |
|---|---|---|---|---|
| DSC (Bulk Reference) | Heat Flow Change | ~10 mg (bulk average) | 85.2 ± 0.5 | Standard protocol; provides global average. |
| Dielectric Spectroscopy | α-Relaxation Time (τ=100 s) | ~50 µL volume | 84.7 ± 1.0 | Directly probes molecular mobility linked to Tg; kinetics of relaxation. |
| Local Thermal Analysis (nano-TA) | Local Softening Temperature | ~100 nm diameter tip contact | 85.5 ± 3.5 (distribution) | Reveals spatial heterogeneity; validates sample homogeneity assumed by DSC/DES. |
Interpretation: Close agreement between the bulk-average DSC Tg and the molecularly-focused DES Tg,diel confirms the global transition. The mean Tg,LTA aligns with these values, while its distribution width provides critical evidence of sample homogeneity, strengthening the validity of the primary DSC protocol in the thesis.
5. Visualization of the Cross-Validation Workflow
Title: Cross-Validation Workflow for Tg Analysis
Title: Decision Logic for Resolving DSC Ambiguities
Benchmarking Against Literature and Reference Materials (e.g., USP standards)
Within the broader thesis on establishing a robust, standardized Differential Scanning Calorimetry (DSC) protocol for measuring the glass transition temperature (Tg) of amorphous solid dispersions (ASDs) in pharmaceutical development, benchmarking against established literature and certified reference materials (CRMs) is paramount. This process validates the accuracy, precision, and reliability of the developed in-house method. Pharmacopeial standards, particularly those from the United States Pharmacopeia (USP), provide a critical foundation for this benchmarking exercise, ensuring data integrity and regulatory compliance.
Application Notes: The Role of USP Standards in DSC-Tg Method Validation
USP general chapter <891> "Thermal Analysis" provides foundational principles. For Tg measurement, specific monographs for drugs or excipients in amorphous form may list Tg or recommend thermal methods. The primary application of USP standards in this context is through chemical CRMs with certified melting points, which are used for temperature calibration of the DSC. Indium is the most common, but tin, zinc, and lead are also used per USP guidance. Accurate temperature calibration is the first critical step in ensuring Tg measurements are comparable to literature values.
Key Application Points:
- Method Verification: Use USP-calibrated DSC to measure Tg of a well-characterized model system (e.g., Polyvinylpyrrolidone K30, Sucrose). Compare results to peer-reviewed literature values (e.g., PVP K30 Tg ~160-170°C; Sucrose Tg ~62-70°C) to verify the protocol's accuracy.
- System Suitability: Establish system suitability tests using CRMs before each batch of Tg analyses. A calibrated DSC must recover the melting point of Indium within ±0.5°C.
- Inter-laboratory Comparison: Benchmarking enables meaningful comparison of Tg data generated across different laboratories, instruments, and operators, which is essential for collaborative research and quality-by-design (QbD) submissions.
Quantitative Data from Literature and Standards
Table 1: Common DSC Calibration Standards (USP-recommended)
| Reference Material | Certified Melting Point (°C) | Purity (%) | Primary Use in Tg Protocol |
|---|---|---|---|
| Indium | 156.60 ± 0.50 | ≥99.999 | Primary temperature & enthalpy calibration |
| Tin | 231.93 ± 0.50 | ≥99.999 | Secondary temperature calibration |
| Zinc | 419.53 ± 0.50 | ≥99.999 | High-temperature calibration check |
| Lead | 327.46 ± 0.50 | ≥99.999 | Secondary temperature calibration |
Table 2: Literature Tg Values for Model Amorphous Systems
| Model System | Literature Tg Range (°C) | Key Literature Source | Notes on Measurement Conditions |
|---|---|---|---|
| Polyvinylpyrrolidone (PVP K30) | 160 - 172 | Surana et al., Pharm. Dev. Technol., 2004 | 10°C/min, N₂ purge, second heat |
| Sucrose | 62 - 70 | Roos & Karel, Biotechnol. Prog., 1991 | 5-20°C/min, dry sample, first heat |
| Polyvinylpyrrolidone-vinyl acetate (PVPVA64) | 106 - 108 | Six et al., J. Pharm. Sci., 2005 | 10°C/min, sealed pan, second heat |
| Felodipine (amorphous) | 45 - 48 | Baird & Taylor, J. Pharm. Sci., 2012 | 10°C/min, dry N₂, first heat |
Experimental Protocols
Protocol 1: DSC Temperature Calibration Using USP-Grade Indium
Objective: To calibrate the DSC temperature axis to within ±0.5°C of the certified value for Indium, as a prerequisite for accurate Tg measurement.
Materials:
- DSC instrument with refrigerated cooling system.
- Standard aluminum Tzero pans and lids (or instrument-specific).
- Analytical balance (0.01 mg sensitivity).
- USP-grade Indium reference standard (≥99.999% purity).
- Hermetic sealing press.
Methodology:
- Preparation: Weigh 5-10 mg of indium using tweezers. Place it in the center of a tared DSC pan. Flatten the metal.
- Sealing: Hermetically seal the pan using the press according to the manufacturer's instructions.
- Instrument Setup: Place the sealed pan in the DSC sample furnace. Leave the reference furnace empty or with an empty sealed pan. Purge the cell with dry nitrogen at 50 mL/min.
- Method Programming: Create a thermal method:
- Equilibrate at 100°C.
- Isotherm for 2 min.
- Ramp from 100°C to 180°C at 10°C/min.
- Run: Execute the method.
- Analysis & Calibration: Determine the onset temperature of the indium melt endotherm. Apply a temperature offset in the instrument software so that the measured onset equals 156.60°C. Save this calibration file.
Protocol 2: Benchmarking Tg Protocol Using Amorphous Sucrose
Objective: To validate the developed DSC Tg measurement protocol by determining the Tg of amorphous sucrose and comparing it to the accepted literature range (62-70°C).
Materials:
- DSC calibrated per Protocol 1.
- Standard aluminum DSC pans (non-hermetic, with pinhole lids).
- Analytical balance.
- Crystalline sucrose (analytical grade).
- Desiccator with phosphorus pentoxide (P₂O₅).
Methodology:
- Sample Preparation (Amorphization): Weigh 3-5 mg of crystalline sucrose into an open DSC pan. Heat the sample in the DSC using a method that melts and quenches it: equilibrate at 25°C, ramp at 50°C/min to 200°C (above sucrose melt), hold for 1 min, then rapidly cool to -50°C at the maximum instrument cooling rate (e.g., 100°C/min).
- Tg Measurement: Immediately run the Tg measurement method on the now-amorphous sample:
- Equilibrate at -50°C.
- Isotherm for 5 min to stabilize.
- Ramp from -50°C to 150°C at 10°C/min under N₂ purge (50 mL/min).
- Data Analysis: Analyze the resulting heat flow curve. Use the midpoint (half-height) method to determine the Tg value from the step transition in the baseline.
- Benchmarking: Compare the measured Tg value to the range in Table 2. A result between 62-70°C, typically ~67°C at 10°C/min, validates the protocol's accuracy for low-Tg carbohydrates. Document any deviation and investigate (e.g., moisture content, scanning rate).
Visualization: Benchmarking Workflow
Title: DSC Tg Method Validation and Benchmarking Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for DSC-Tg Benchmarking Experiments
| Item | Function in Benchmarking | Example/Specification |
|---|---|---|
| USP-Grade Calibration Standards | Provide traceable, certified melting points for instrument calibration, ensuring temperature accuracy. | Indium, Tin, Zinc (≥99.999% purity). |
| Model Amorphous Systems | Well-characterized materials with published Tg values used to verify the entire measurement protocol. | Amorphous Sucrose, PVP K30, PVPVA64. |
| High-Purity Inert Gas | Prevents oxidative degradation during heating and ensures stable baseline. Dry gas is critical for hygroscopic samples. | Nitrogen or Argon, 99.999% purity, with inline moisture trap. |
| Standard DSC Pan Systems | Provide consistent thermal contact and mass for reproducible results. Type depends on sample. | Tzero Aluminum Hermetic pans (for liquids/volatiles), standard pans with pinhole lids (for solids). |
| Microbalance | Accurately measures small sample masses (1-10 mg) critical for optimal thermal response and resolution of Tg. | Capacity 0.01 mg (10 µg). |
| Desiccant | Used to store and prepare hygroscopic samples (most ASDs) to control plasticizing effects of moisture on Tg. | Phosphorus pentoxide (P₂O₅) or molecular sieves in a desiccator. |
| Hermetic Sealing Press | Ensures complete encapsulation of samples, preventing mass loss and controlling sample environment. | Manufacturer-specific press for DSC pans. |
Within a comprehensive thesis on Differential Scanning Calorimetry (DSC) protocols for measuring the glass transition temperature (Tg), understanding the role of plasticizers, particularly water, is paramount. Water acts as a potent low-molecular-weight plasticizer for many amorphous materials, including polymers and biopharmaceutical formulations, significantly depressing the Tg. This depression can critically impact the physical stability, processing, and storage conditions of materials like amorphous solid dispersions and lyophilized proteins. Accurate modeling of this relationship is essential for predictive stability studies. The Gordon-Taylor equation provides a fundamental thermodynamic framework for modeling the plasticizing effect of water on Tg, enabling researchers to predict Tg as a function of moisture content.
Core Theory: The Gordon-Taylor Equation
The Gordon-Taylor equation is a semi-empirical model used to predict the glass transition temperature of a mixture (Tg,mix) based on the Tg of the individual components and their weight fractions.
Equation: Tg,mix = (w1 * Tg1 + K * w2 * Tg2) / (w1 + K * w2)
Where:
- w1, w2 = weight fractions of components 1 and 2 (e.g., polymer/drug and water).
- Tg1, Tg2 = glass transition temperatures (in Kelvin) of the pure components.
- K = a fitting constant often related to the strength of interaction and the difference in free volume between the components. It can be approximated by K ≈ (ρ1 * Δα2) / (ρ2 * Δα1), where ρ is density and Δα is the change in thermal expansion coefficient at Tg.
Application Notes: Data Modeling and Interpretation
Table 1: Example Data for Tg Depression of a Model Polymer by Water
| Water Content (% w/w) | Experimental Tg (°C) | Gordon-Taylor Prediction (°C) | Deviation (°C) |
|---|---|---|---|
| 0.0 | 105.0 | 105.0 | 0.0 |
| 1.5 | 92.5 | 91.8 | +0.7 |
| 3.0 | 79.0 | 78.9 | +0.1 |
| 5.0 | 60.2 | 61.5 | -1.3 |
| 7.0 | 45.1 | 44.3 | +0.8 |
Assumptions for prediction: Tg1 = 105°C, Tg2 = -135°C, K = 5.2
Key Insights:
- Non-Linear Depression: The depression of Tg is highly non-linear; small amounts of water at low moisture levels cause a significant initial decrease.
- Parameter K: The value of K is critical. A higher K indicates a weaker plasticizing effect of the diluent (water) for that specific system.
- Predictive Power: When fitted correctly, the Gordon-Taylor equation provides excellent predictive capability for Tg over a range of moisture contents, crucial for determining critical storage conditions (e.g., below Tg - 50°C for stability).
Experimental Protocols
Protocol 4.1: DSC Measurement of Tg for Humidified Samples
Objective: To determine the glass transition temperature of a material equilibrated at various relative humidities (RH). Materials: See "The Scientist's Toolkit" below. Procedure:
- Sample Preparation:
- Place approximately 5-10 mg of the dry amorphous material (e.g., polymer, lyophilized protein) into multiple tared, open DSC pans.
- Transfer pans to controlled humidity desiccators containing saturated salt solutions (e.g., LiCl for 11% RH, MgCl2 for 33% RH, NaCl for 75% RH).
- Store desiccators at constant temperature (e.g., 25°C) for a minimum of 7 days to ensure sorption equilibrium.
- Weight Measurement:
- Rapidly remove pans from desiccators, seal immediately with a hermetic lid, and record the precise sample weight using a microbalance.
- Calculate the final moisture content (% w/w).
- DSC Analysis:
- Load sealed pan into the DSC furnace. Use an empty sealed pan as a reference.
- Method: Equilibrate at -50°C. Ramp at 10°C/min to a temperature above the expected Tg. Use a nitrogen purge gas (50 mL/min).
- Perform triplicate runs for each humidity condition.
- Data Analysis:
- Analyze the thermogram using the instrument software. Take Tg as the midpoint of the heat capacity change.
- Plot experimental Tg (in Kelvin) vs. water weight fraction.
Protocol 4.2: Fitting Data to the Gordon-Taylor Equation
Objective: To obtain the fitting parameter K and validate the model. Procedure:
- Transform all Tg data to Kelvin (K = °C + 273.15).
- Using statistical software (e.g., Origin, Prism), fit the data (Tg,mix vs. w_water) to the Gordon-Taylor equation using non-linear regression.
- Fix Tg1 (dry polymer) and Tg2 (water, ~138 K) as constants during fitting, allowing K to be the sole fitting parameter.
- Assess the goodness of fit using R² and residual plots.
- Use the fitted equation to predict Tg at untested moisture levels.
Visualizations
Title: Experimental Workflow for Tg vs. Moisture Content Study
Title: Inputs and Output of the Gordon-Taylor Model
The Scientist's Toolkit
Table 2: Essential Research Reagents and Materials
| Item | Function / Explanation |
|---|---|
| Hermetic DSC Pans & Lids | Sealed, volatile-resistant pans to prevent moisture loss during DSC run, ensuring the measured Tg corresponds to the prepared water content. |
| Saturated Salt Solutions | Provides a constant relative humidity (RH) environment in a desiccator for sample equilibration (e.g., K₂CO₃ for 43% RH, Mg(NO₃)₂ for 53% RH). |
| High-Precision Microbalance | Accurately measures small changes in sample mass (μg level) to determine absorbed water content (% w/w) after humidity equilibration. |
| Nitrogen Gas Supply | Inert purge gas for the DSC cell to prevent oxidative degradation and ensure a stable, moisture-free atmosphere during measurement. |
| Statistical Software | Required for non-linear regression fitting of experimental Tg data to the Gordon-Taylor equation to derive the parameter K. |
| Standard Reference Materials | Indium, Zinc for calibration of DSC temperature and enthalpy scales, ensuring accurate and reproducible Tg measurements. |
Importance of Tg in Regulatory Filings and Quality-by-Design (QbD) Frameworks
Within the context of a broader thesis on Differential Scanning Calorimetry (DSC) protocol development, the measurement of the glass transition temperature (Tg) is established as a critical quality attribute (CQA) for amorphous solid dispersions, polymeric excipients, and biopharmaceutical formulations. Its determination is integral to both regulatory submissions and the systematic QbD approach, linking material science directly to drug product stability, performance, and manufacturability.
Regulatory Significance of Tg
Tg data is a mandated component of regulatory filings (e.g., FDA, EMA, ICH) for products containing amorphous materials. It directly informs the control strategy by defining storage conditions, packaging requirements, and shelf-life.
| Regulatory Body/Guideline | Reference Code | Relevance to Tg | Key Implication |
|---|---|---|---|
| International Council for Harmonisation (ICH) | ICH Q6A | Specification for Polymorphs | Tg may serve as an indirect test for physical state. |
| International Council for Harmonisation (ICH) | ICH Q8(R2) | Pharmaceutical Development | Tg is a potential CQA underpinning design space. |
| International Council for Harmonisation (ICH) | ICH Q1A(R2) | Stability Testing | Tg defines storage condition limits (e.g., below Tg-50°C). |
| U.S. Food and Drug Administration (FDA) | PAT Guidance | Process Analytical Technology | In-line monitoring of Tg during processes like drying. |
| European Medicines Agency (EMA) | EMA/CHMP/493702/2018 | Quality of Oral Modified Release Products | Critical for matrix polymer performance. |
Tg in the QbD Framework
In QbD, Tg is utilized to establish relationships between material attributes, process parameters, and the CQAs of the drug product.
Diagram 1: Tg's Role in the QbD Workflow
Title: Tg as a Link Between CMAs, CPPs, and CQAs in QbD
Table 2: Tg as a CMA and Its Impact on Product CQAs
| Drug Product System | Tg as CMA | Linked CQA | Target Relationship |
|---|---|---|---|
| Amorphous Solid Dispersion | Tg of the dispersion | Chemical stability, Dissolution rate | Tg > Storage Temp + 50°C |
| Lyophilized (Freeze-Dried) Product | Tg' or Tg of cake | Reconstitution time, Residual moisture | High Tg ensures cake structure |
| Controlled Release Matrix | Tg of polymer film | Drug release profile | Tg affects polymer mobility & erosion |
| Spray-Dried Intermediate | Tg of powder | Flowability, Compressibility | High Tg reduces particle agglomeration |
Application Notes: DSC Protocol for Tg Determination
A robust, validated DSC protocol is fundamental for generating reliable Tg data for regulatory and QbD purposes.
Application Note 1: Method Development for Amorphous Dispersions
Objective: To determine the Tg of a spray-dried amorphous dispersion with high precision to define the product's storage condition design space. Challenge: Separating the Tg signal from relaxation endotherms and moisture effects. Protocol Solution:
- Sample Preparation: Dry powder in a desiccator (P₂O₅) for 24h. Hermetically seal sample in a pierced lid crucible.
- DSC Method:
- Temperature Range: 25°C to 150°C (above predicted Tg).
- Heating Rate: 10°C/min (standard). Also run at 20°C/min to shift relaxation away from Tg.
- Cooling Rate: 50°C/min.
- Atmosphere: Dry N₂ at 50 mL/min.
- Cycle: First heat (analyze), cool, second heat (definitive Tg). Report Tg from the second heating scan to erase thermal history.
- Tg Analysis: Use midpoint (half-height) method for reporting. Onset and endpoint values are recorded for design space boundaries.
Application Note 2: Tg' in Lyophilization Formulation Screening
Objective: To identify the critical formulation parameter Tg' for cycle development. Challenge: Measuring the glass transition of the maximally freeze-concentrated solute. Protocol Solution:
- Sample Preparation: Load 10-20 µL of aqueous formulation solution into a high-volume DSC pan.
- DSC Method:
- Cooling: Equilibrate at 20°C, cool to -80°C at 10°C/min.
- Heating: Heat from -80°C to +20°C at 5°C/min.
- Analysis: Identify the onset of the endothermic step change in the reversing heat flow signal during warming. This onset temperature is Tg'.
Detailed Experimental Protocols
Protocol 1: Standard DSC Tg Measurement for Regulatory Filing
Title: Determination of Glass Transition Temperature by Differential Scanning Calorimetry SOP Code: DSC-TG-001 Scope: This protocol describes the procedure for measuring the Tg of pharmaceutical solids using a power-compensated or heat-flux DSC. Materials: See Scientist's Toolkit below. Method:
- Calibration: Calibrate the DSC for temperature and enthalpy using indium and zinc standards.
- Baseline Recording: Run an empty pan vs. reference pan using the method parameters. Save as baseline.
- Sample Preparation:
- Weigh 5-10 mg of sample accurately into a tared DSC pan.
- For hygroscopic samples, perform in a dry box.
- Hermetically seal the pan with a lid pierced with a laser or pinhole.
- Instrument Parameters:
- Purge Gas: Nitrogen, 50 mL/min.
- Heating Rate: 10°C/min.
- Temperature Range: Start at least 50°C below expected Tg, end 30°C above.
- Run Sequence:
- Equilibrate at start temperature.
- First Heat: Heat at 10°C/min to endpoint. (Observes history).
- Cool: Rapid cool at 50°C/min to start temperature.
- Second Heat: Repeat heating scan. This scan provides the reported Tg.
- Data Analysis:
- Subtract the saved baseline from the sample curve.
- On the second heat curve, identify the glass transition region.
- Use the software tangent method to determine Tg-onset, Tg-midpoint, and Tg-endpoint.
- Report Tg-midpoint as the primary value (in °C). Document ΔCp (change in heat capacity).
Protocol 2: Modulated DSC (mDSC) for Complex Systems
Title: Separation of Tg from Overlapping Events using mDSC SOP Code: mDSC-TG-001 Scope: For separating the reversible Tg from non-reversible events (e.g., enthalpy relaxation, evaporation, decomposition). Method:
- Sample Preparation: As per Protocol 1.
- Instrument Parameters:
- Underlying Heating Rate: 2°C/min.
- Modulation Amplitude: ±0.5°C.
- Modulation Period: 60 seconds.
- Purge Gas: Nitrogen, 50 mL/min.
- Run Sequence: Single heat from below to above Tg.
- Data Analysis:
- Analyze the Reversing Heat Flow signal.
- The Tg is identified as a step change in this signal, free from overlapping endothermic peaks present in the Total Heat Flow.
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions and Materials
| Item | Function/Brand Example (Illustrative) | Critical Use in Tg Protocol |
|---|---|---|
| Differential Scanning Calorimeter | e.g., TA Instruments Q Series, Mettler Toledo DSC 3 | Primary instrument for thermal analysis. |
| Hermetic Sealed DSC Pans & Lids | Tzero pans (TA), 40µL crucibles (Mettler) | Standard containment for most solids. |
| High-Volume Hermetic DSC Pans | For liquids or solutions (e.g., lyophilization studies). | Prevents evaporation during run. |
| Dielectric Sealant | e.g., Silicon-based grease (for humidity control) | For sealing pans in humidity studies. |
| Ultra-Pure Calibration Standards | Indium (Tm=156.6°C), Zinc (Tm=419.5°C), NIST-traceable. | Mandatory temperature/enthalpy calibration. |
| Desiccant | e.g., Phosphorus pentoxide (P₂O₅) | Drying hygroscopic samples pre-analysis. |
| Dry Box/Glove Box | Controlled atmosphere enclosure (<1% RH). | Sample preparation for moisture-sensitive APIs. |
| Microbalance | Capacity 1-5 mg, readability 0.001 mg. | Accurate sample weighing. |
| mDSC Software Module | Optional but recommended for complex systems. | Deconvolutes reversing/non-reversing heat flow. |
Data Presentation for Regulatory & QbD
| Formulation Batch | Process Parameter (Spray Dry Inlet Temp) | Tg Midpoint (°C) ± SD (n=3) | ΔCp (J/g°C) | Related Stability Outcome (40°C/75% RH, 3M) |
|---|---|---|---|---|
| F-01 | 120°C | 65.2 ± 0.5 | 0.35 | Failed: >2% degradation, crystallization observed. |
| F-02 | 150°C | 78.5 ± 0.3 | 0.31 | Passed: <0.5% degradation, amorphous maintained. |
| F-03 | 180°C | 80.1 ± 0.4 | 0.30 | Passed: <0.5% degradation, amorphous maintained. |
| Design Space Limit | 140-170°C | >75°C | N/A | Proven acceptable range for stability. |
Diagram 2: DSC Tg Data Informs Control Strategy
Title: From DSC Data to Control Strategy and Filing
The accurate determination of Tg via a robust DSC protocol is non-negotiable in modern pharmaceutical development. It serves as a pivotal link between the molecular characteristics of a formulation and its macroscopic performance, providing a scientific basis for regulatory claims and forming the backbone of a proactive QbD control strategy. Within a thesis on DSC methodology, this underscores the transition from a technique of mere characterization to one of fundamental risk mitigation and quality assurance.
Conclusion
The accurate measurement of Tg via DSC is a cornerstone of modern pharmaceutical development for amorphous solid dispersions, biologics, and lyophilized products. A robust protocol, as outlined, begins with a deep understanding of the material's physics, follows a meticulous experimental method, anticipates and troubleshoots analytical challenges, and is validated with complementary techniques. Mastery of DSC for Tg empowers scientists to predict physical stability, rationalize formulation choices, and design reliable manufacturing processes. Future directions involve integrating high-throughput DSC with computational modeling for predictive stability and the development of standardized, harmonized protocols for global regulatory acceptance, ultimately accelerating the delivery of stable, effective medicines to patients.